What is the general name of the organic compounds containing the
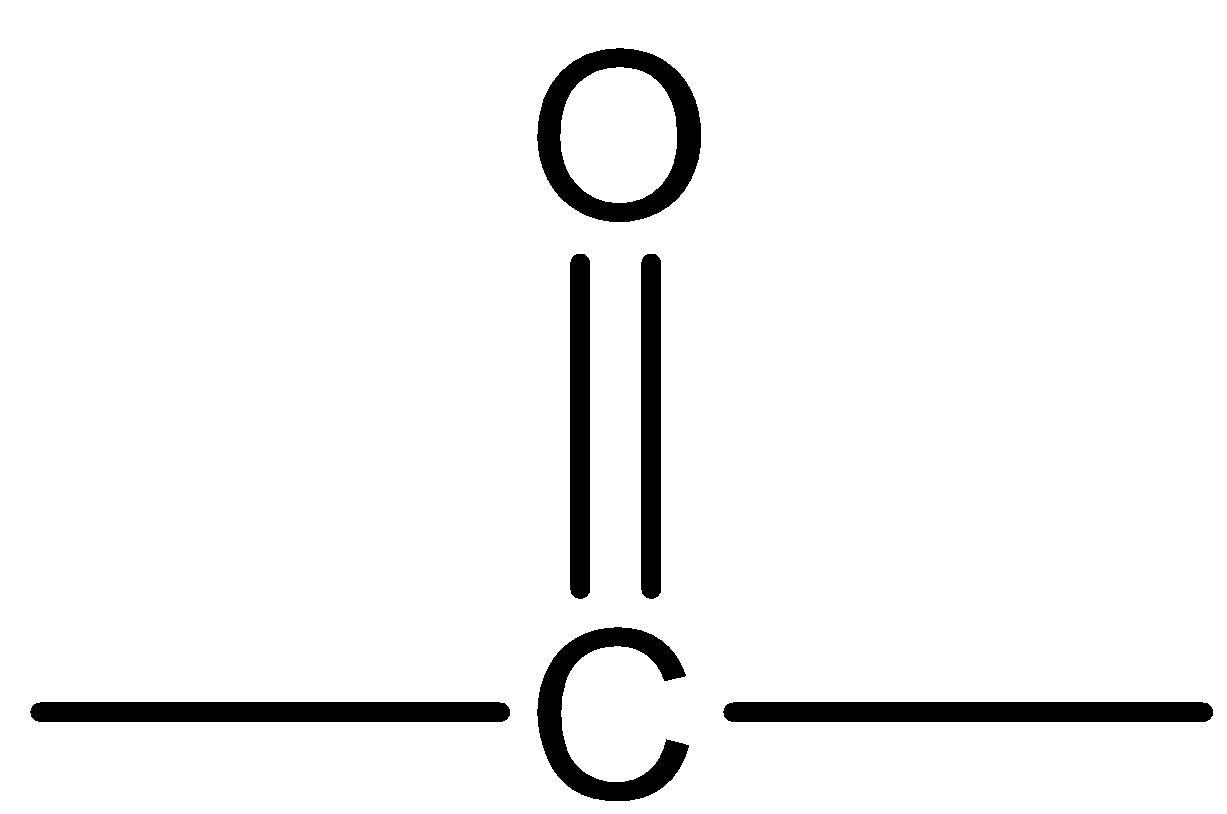 group?
group?

Answer
529.5k+ views
Hint: As we know that an organic compound has both common names and IUPAC names. The common name is also known as general names. Each functional group has its own general names. And the IUPAC nomenclature is a chemical nomenclature which is used for the naming of organic compounds by following some rules and regulations. And it is proposed by the International Union of Pure and Applied Chemistry. The name is different for each functional group.
Complete answer:
The general name of the given organic compound containing the
The group is known as carbonyl or ketone. The structure of the functional group present in the ketone is\[{R_2}C = O\]. Here, R may be the substituents which contain carbon atoms and it contains carbon – oxygen double bonds. The IUPAC name of a ketone should contain –one or –oxo. The general formula of a period is \[{C_n}{H_{2n}}O\]. Where, n is equal to the number of carbon atoms.
The ketone has many uses. The ketone is used as a solvent for the preparation of synthetic fibers and plastics. And it is also used for the procedures like chemical peeling, medical purposes and it is used for removing the nail polish.
Note:
We need to know that the functional groups are the structural units present in the organic compounds. It has a specific bonding arrangement in a particular atom. For example, the alkene containing carbon – carbon double bond and alkyne containing the carbon – carbon single bond. And the alcohol containing the hydroxyl functional groups, aldehyde and ketone containing the carbonyl groups.
Complete answer:
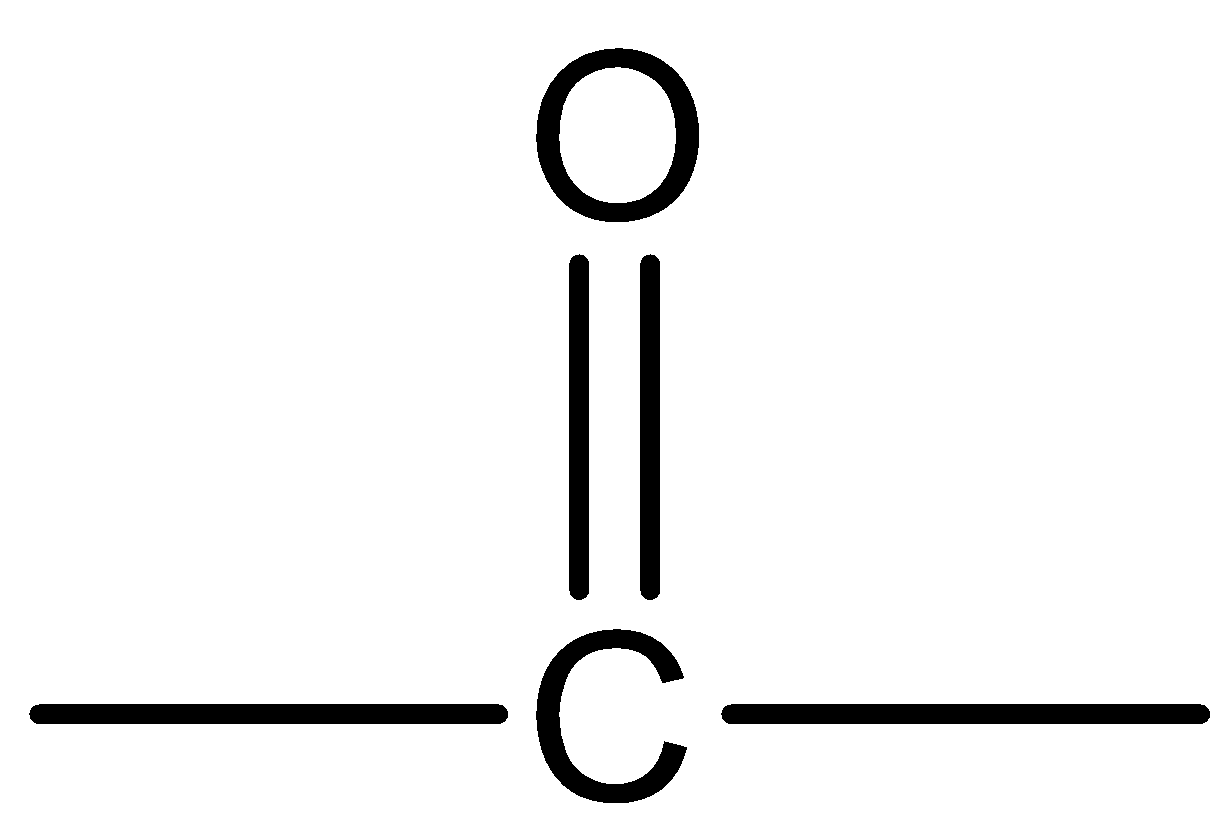
The general name of the given organic compound containing the

The group is known as carbonyl or ketone. The structure of the functional group present in the ketone is\[{R_2}C = O\]. Here, R may be the substituents which contain carbon atoms and it contains carbon – oxygen double bonds. The IUPAC name of a ketone should contain –one or –oxo. The general formula of a period is \[{C_n}{H_{2n}}O\]. Where, n is equal to the number of carbon atoms.
The ketone has many uses. The ketone is used as a solvent for the preparation of synthetic fibers and plastics. And it is also used for the procedures like chemical peeling, medical purposes and it is used for removing the nail polish.
Note:
We need to know that the functional groups are the structural units present in the organic compounds. It has a specific bonding arrangement in a particular atom. For example, the alkene containing carbon – carbon double bond and alkyne containing the carbon – carbon single bond. And the alcohol containing the hydroxyl functional groups, aldehyde and ketone containing the carbonyl groups.
Recently Updated Pages
Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Master Class 11 Chemistry: Engaging Questions & Answers for Success

Master Class 11 Biology: Engaging Questions & Answers for Success

Master Class 11 Physics: Engaging Questions & Answers for Success

Trending doubts
Differentiate between an exothermic and an endothermic class 11 chemistry CBSE

One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

There are 720 permutations of the digits 1 2 3 4 5 class 11 maths CBSE

Draw a diagram of a plant cell and label at least eight class 11 biology CBSE

Two of the body parts which do not appear in MRI are class 11 biology CBSE




