For the complex ion \[{\left[ {{\text{Fe}}{{\left( {{\text{CN}}} \right)}_6}} \right]^{3 - }}\] state:
(i) The geometry of the ion.
(ii) The magnetic property of the ion.
Answer
599.1k+ views
Hint: Six strong field ligands are attached to the central metal atom ${\text{Fe}}$. If all the electrons in the complex ion are paired it is diamagnetic in nature. If the complex ion has an unpaired electron, it is paramagnetic in nature.
Step by step answer: Determine the geometry of the ion as follows:
In the complex ion \[{\left[ {{\text{Fe}}{{\left( {{\text{CN}}} \right)}_6}} \right]^{3 - }}\], the ligand is ${\text{C}}{{\text{N}}^ - }$. The ligand ${\text{C}}{{\text{N}}^ - }$ can accept electrons from the ${\text{Fe}}$ metal atom. Thus, ${\text{C}}{{\text{N}}^ - }$ is a strong field ligand.
As ${\text{C}}{{\text{N}}^ - }$ is a strong field ligand. Thus, a low spin complex is formed using the inner ${\text{3d}}$ orbitals of the ${\text{Fe}}$ metal atom.
There are six ${\text{C}}{{\text{N}}^ - }$ ligands. Thus, the geometry of the complex ion \[{\left[ {{\text{Fe}}{{\left( {{\text{CN}}} \right)}_6}} \right]^{3 - }}\] is octahedral.
Determine the magnetic property of the ion as follows:
Determine the oxidation number (ON) of the central metal atom ${\text{Fe}}$ as follows:
${\text{ON of Fe + 6}} \times {\text{ON of C}}{{\text{N}}^ - } = {\text{Charge of the complex ion}}$
Substitute $\left( { - 1} \right)$ for the oxidation number of ${\text{C}}{{\text{N}}^ - }$, $ - 3$ for the charge on the complex. Thus,
${\text{ON of Fe + 6}} \times \left( { - 1} \right) = - 3$
${\text{ON of Fe}} = - 3 + 6$
${\text{ON of Fe}} = + 3$
Thus, the oxidation number (ON) of the central metal atom ${\text{Fe}}$ is $ + 3$.
Determine the electronic configuration as follows:
The atomic number of iron is ${\text{26}}$. The electronic configuration of the ${\text{Fe}}$ metal is $1{s^2}\,2{s^2}\,2{p^6}\,3{s^2}\,3{p^6}\,3{d^6}\,4{s^2}$.
The ${\text{Fe}}$ metal is in $ + 3$ state. Thus, ${\text{Fe}}$ metal loses three electrons, two from the ${\text{4s}}$ orbital and one from the ${\text{3d}}$ orbital. Thus, the electronic configuration of ${\text{Fe}}$ in $ + 3$ state is $1{s^2}\,2{s^2}\,2{p^6}\,3{s^2}\,3{p^6}\,3{d^5}$.
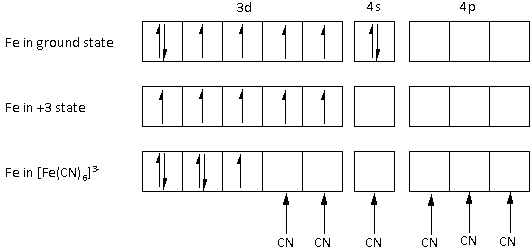
After pairing, the complex ion \[{\left[ {{\text{Fe}}{{\left( {{\text{CN}}} \right)}_6}} \right]^{3 - }}\] has one unpaired electron. Thus, the complex ion \[{\left[ {{\text{Fe}}{{\left( {{\text{CN}}} \right)}_6}} \right]^{3 - }}\] is paramagnetic.
Thus, the magnetic property of the ion \[{\left[ {{\text{Fe}}{{\left( {{\text{CN}}} \right)}_6}} \right]^{3 - }}\] is paramagnetic.
Note: The strong field ligands form low spin complexes and prefer the pairing of electrons. The weak field ligands form high spin complexes and do not prefer the pairing of electrons. The charge on the ${\text{C}}{{\text{N}}^ - }$ ligand is $ - 1$. And the charge on the central metal atom ${\text{Fe}}$ is $ + 3$ and not $ + 2$.
Step by step answer: Determine the geometry of the ion as follows:
In the complex ion \[{\left[ {{\text{Fe}}{{\left( {{\text{CN}}} \right)}_6}} \right]^{3 - }}\], the ligand is ${\text{C}}{{\text{N}}^ - }$. The ligand ${\text{C}}{{\text{N}}^ - }$ can accept electrons from the ${\text{Fe}}$ metal atom. Thus, ${\text{C}}{{\text{N}}^ - }$ is a strong field ligand.
As ${\text{C}}{{\text{N}}^ - }$ is a strong field ligand. Thus, a low spin complex is formed using the inner ${\text{3d}}$ orbitals of the ${\text{Fe}}$ metal atom.
There are six ${\text{C}}{{\text{N}}^ - }$ ligands. Thus, the geometry of the complex ion \[{\left[ {{\text{Fe}}{{\left( {{\text{CN}}} \right)}_6}} \right]^{3 - }}\] is octahedral.
Determine the magnetic property of the ion as follows:
Determine the oxidation number (ON) of the central metal atom ${\text{Fe}}$ as follows:
${\text{ON of Fe + 6}} \times {\text{ON of C}}{{\text{N}}^ - } = {\text{Charge of the complex ion}}$
Substitute $\left( { - 1} \right)$ for the oxidation number of ${\text{C}}{{\text{N}}^ - }$, $ - 3$ for the charge on the complex. Thus,
${\text{ON of Fe + 6}} \times \left( { - 1} \right) = - 3$
${\text{ON of Fe}} = - 3 + 6$
${\text{ON of Fe}} = + 3$
Thus, the oxidation number (ON) of the central metal atom ${\text{Fe}}$ is $ + 3$.
Determine the electronic configuration as follows:
The atomic number of iron is ${\text{26}}$. The electronic configuration of the ${\text{Fe}}$ metal is $1{s^2}\,2{s^2}\,2{p^6}\,3{s^2}\,3{p^6}\,3{d^6}\,4{s^2}$.
The ${\text{Fe}}$ metal is in $ + 3$ state. Thus, ${\text{Fe}}$ metal loses three electrons, two from the ${\text{4s}}$ orbital and one from the ${\text{3d}}$ orbital. Thus, the electronic configuration of ${\text{Fe}}$ in $ + 3$ state is $1{s^2}\,2{s^2}\,2{p^6}\,3{s^2}\,3{p^6}\,3{d^5}$.

After pairing, the complex ion \[{\left[ {{\text{Fe}}{{\left( {{\text{CN}}} \right)}_6}} \right]^{3 - }}\] has one unpaired electron. Thus, the complex ion \[{\left[ {{\text{Fe}}{{\left( {{\text{CN}}} \right)}_6}} \right]^{3 - }}\] is paramagnetic.
Thus, the magnetic property of the ion \[{\left[ {{\text{Fe}}{{\left( {{\text{CN}}} \right)}_6}} \right]^{3 - }}\] is paramagnetic.
Note: The strong field ligands form low spin complexes and prefer the pairing of electrons. The weak field ligands form high spin complexes and do not prefer the pairing of electrons. The charge on the ${\text{C}}{{\text{N}}^ - }$ ligand is $ - 1$. And the charge on the central metal atom ${\text{Fe}}$ is $ + 3$ and not $ + 2$.
Recently Updated Pages
Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Master Class 12 Business Studies: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Explain the structure of megasporangium class 12 biology CBSE

What are the major means of transport Explain each class 12 social science CBSE

How many chromosomes are found in human ovum a 46 b class 12 biology CBSE

The diagram of the section of a maize grain is given class 12 biology CBSE




