Find the number of carbon atoms including the given structure which can have negative change in resonating structure. (The structure with charge repeating are not accepted)

Answer
529.5k+ views
Hint: In organic chemistry, hydrocarbons are an important topic. The hydrocarbons are majorly classified as three groups. There are alkane, alkene and alkyne. The alkane means carbon-carbon single bond. The alkene has a carbon-carbon double bond. The alkyne means carbon-carbon having triple bond in the molecule. In organic chemistry stability of molecules and ions are very important. Carbon ions are classified as two types. There is carbocation and carbanion. The carbon atom which has a positive charge is called carbocation. The carbon atom which has negative charge is called carbocation. In organic chemistry cleavage is split into two types. There are homolytic cleavage and heterolytic cleavage. Ions will happen in heterolytic cleavage. Free radicals will come in Homolytic cleavage.
Complete answer:
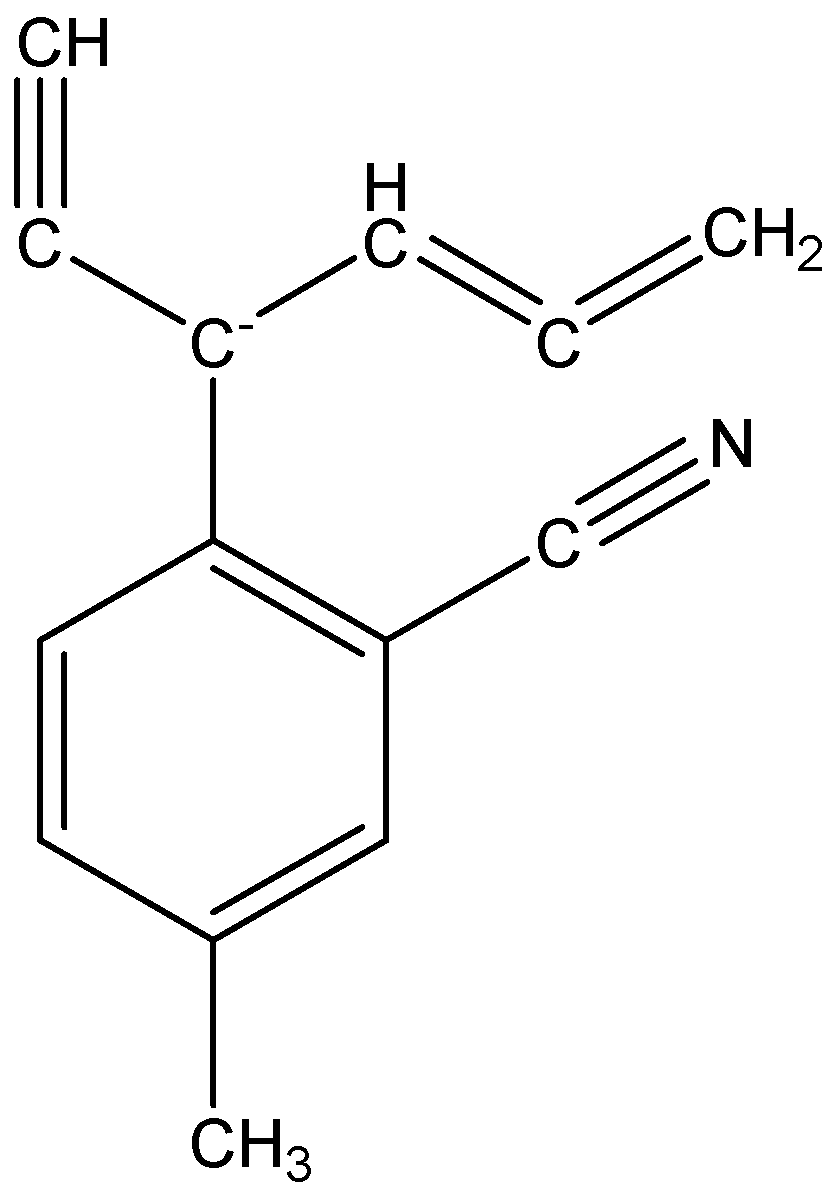
The given structure is
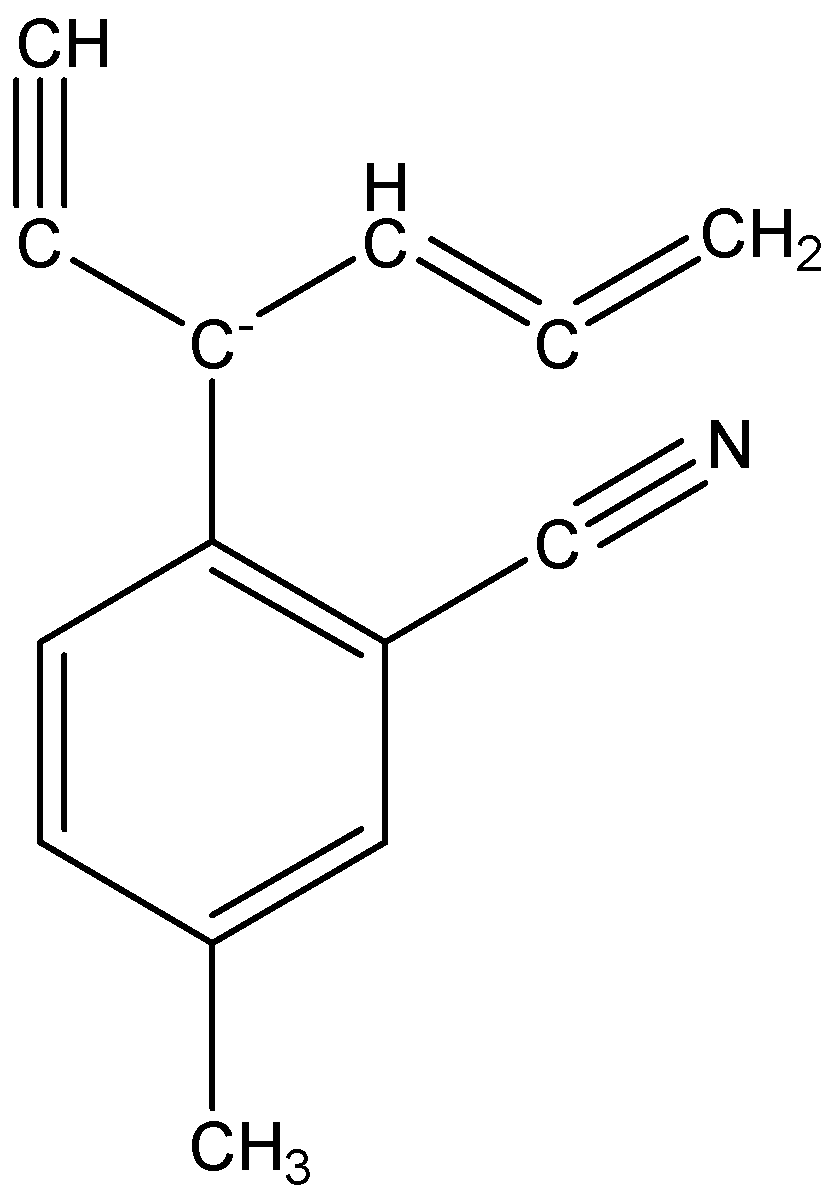
The resonating structure of the given structure which can have negative change in resonating is given below,
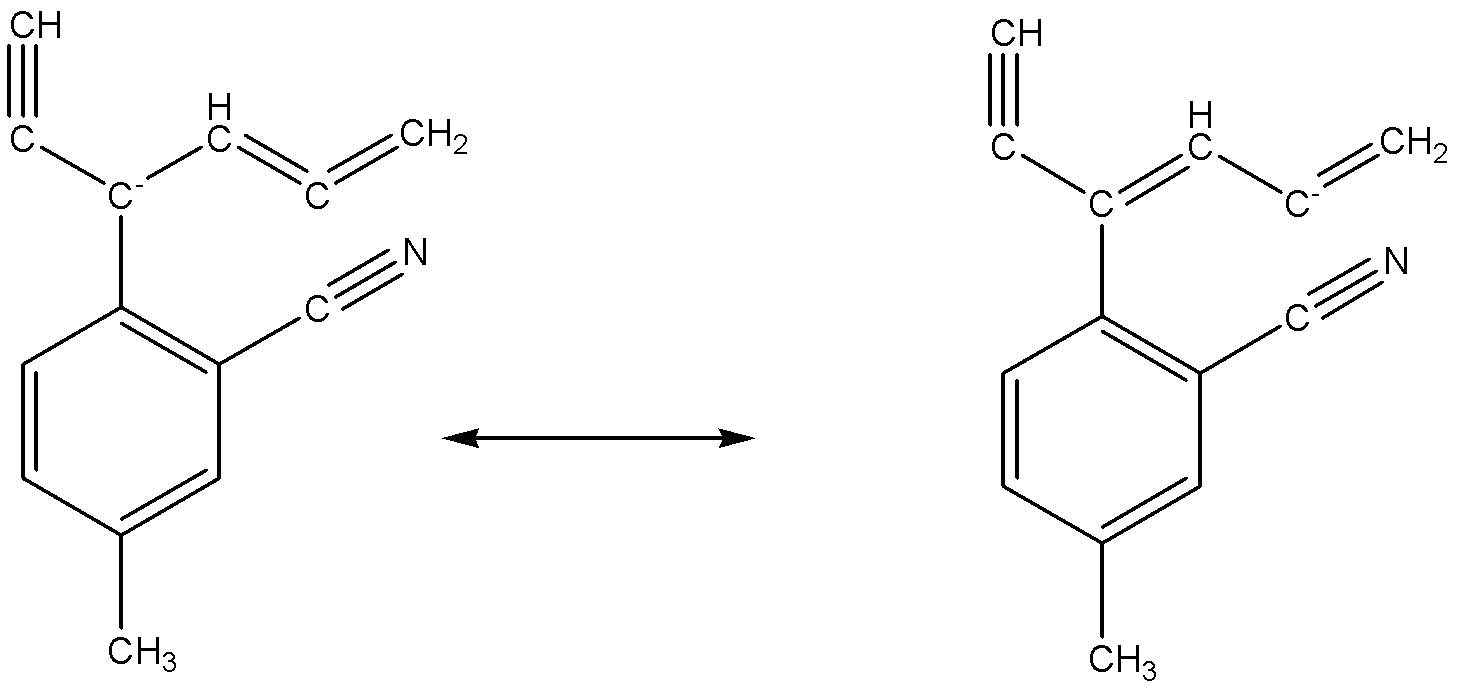
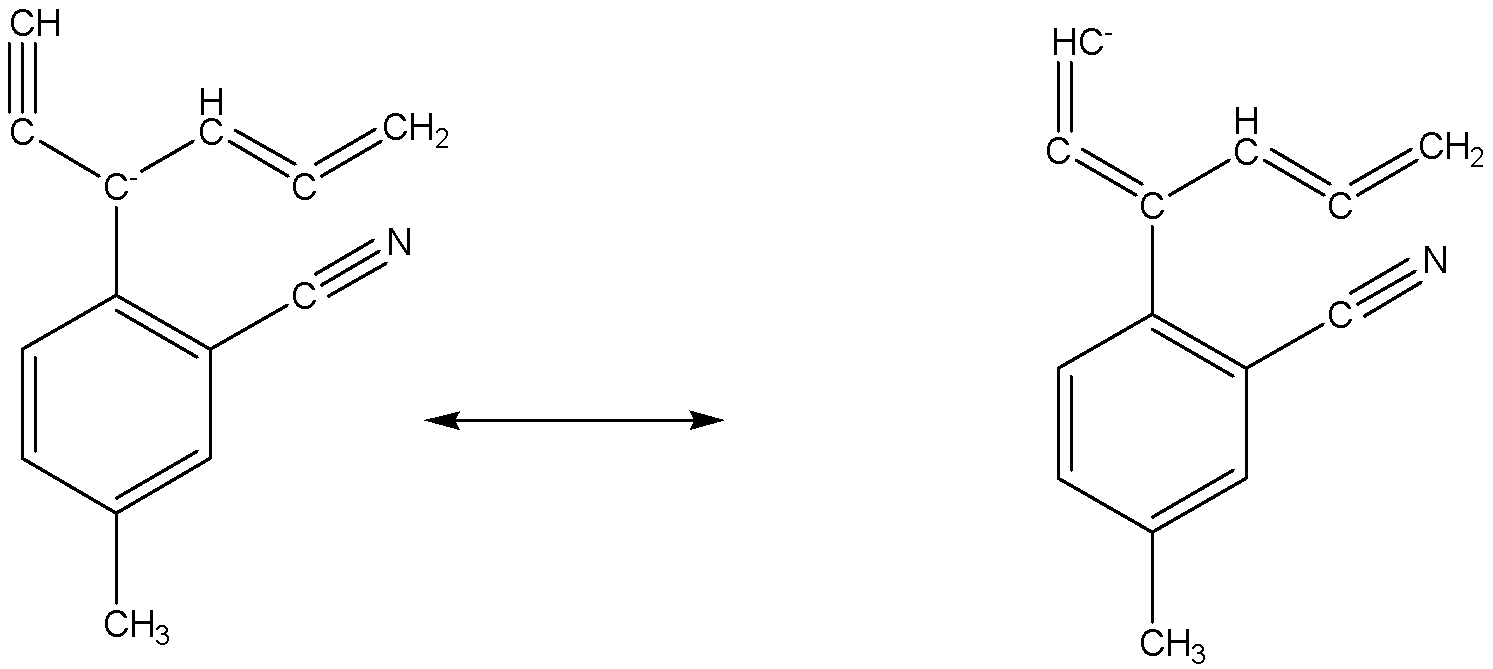
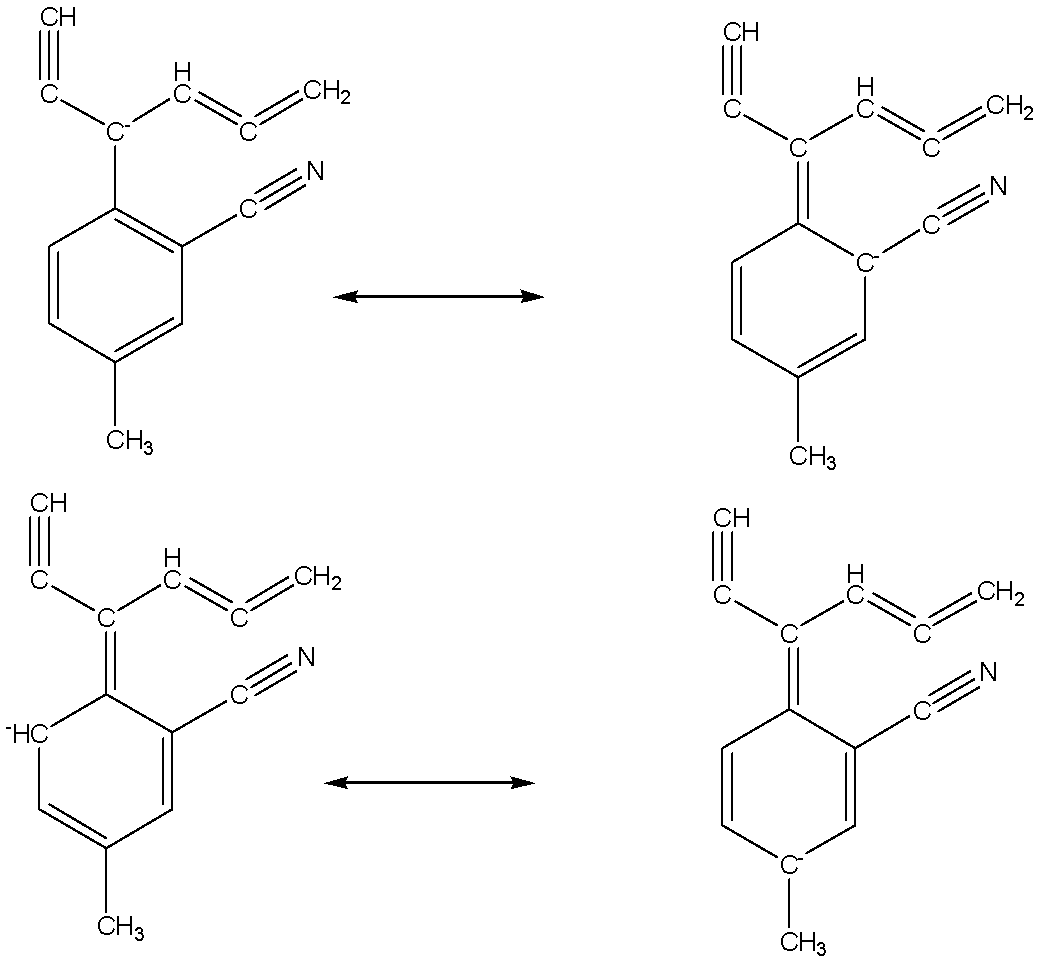
Six number of carbon atoms including the given structure which can have negative change in resonating structures.
Note:
We have to know the conversion of one type of hydrocarbon to another hydrocarbon by oxidation and reduction. The oxidation of alkane to give alkene. The oxidation of alkene to give alkyne. The reduction of alkyne to give alkene. The reduction of alkene to give alkane. It has some general formulas. The general formula of alkane is \[{{\text{C}}_{\text{n}}}{{\text{H}}_{{\text{2n + 2}}}}\]. The general formula of alkene is \[{{\text{C}}_{\text{n}}}{{\text{H}}_{{\text{2n}}}}\]. The general formula of alkyne is \[{{\text{C}}_{\text{n}}}{{\text{H}}_{{\text{2n - 2}}}}\]. The general formula of cycloalkane is \[{{\text{C}}_{\text{n}}}{{\text{H}}_{{\text{2n}}}}\].
Complete answer:
The given structure is

The resonating structure of the given structure which can have negative change in resonating is given below,



Six number of carbon atoms including the given structure which can have negative change in resonating structures.
Note:
We have to know the conversion of one type of hydrocarbon to another hydrocarbon by oxidation and reduction. The oxidation of alkane to give alkene. The oxidation of alkene to give alkyne. The reduction of alkyne to give alkene. The reduction of alkene to give alkane. It has some general formulas. The general formula of alkane is \[{{\text{C}}_{\text{n}}}{{\text{H}}_{{\text{2n + 2}}}}\]. The general formula of alkene is \[{{\text{C}}_{\text{n}}}{{\text{H}}_{{\text{2n}}}}\]. The general formula of alkyne is \[{{\text{C}}_{\text{n}}}{{\text{H}}_{{\text{2n - 2}}}}\]. The general formula of cycloalkane is \[{{\text{C}}_{\text{n}}}{{\text{H}}_{{\text{2n}}}}\].
Recently Updated Pages
Three beakers labelled as A B and C each containing 25 mL of water were taken A small amount of NaOH anhydrous CuSO4 and NaCl were added to the beakers A B and C respectively It was observed that there was an increase in the temperature of the solutions contained in beakers A and B whereas in case of beaker C the temperature of the solution falls Which one of the following statements isarecorrect i In beakers A and B exothermic process has occurred ii In beakers A and B endothermic process has occurred iii In beaker C exothermic process has occurred iv In beaker C endothermic process has occurred

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 Physics: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Master Class 11 Economics: Engaging Questions & Answers for Success

Master Class 11 Computer Science: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

There are 720 permutations of the digits 1 2 3 4 5 class 11 maths CBSE

State and prove Bernoullis theorem class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

1 Quintal is equal to a 110 kg b 10 kg c 100kg d 1000 class 11 physics CBSE

Discuss the various forms of bacteria class 11 biology CBSE




