How can we find the charge of a particular compound like ${\text{N}}{{\text{H}}_{\text{3}}}$…. as I want to take out the oxidation state of ${\left( {{\text{Ni}}{{\left( {{\text{N}}{{\text{H}}_{\text{3}}}} \right)}_{\text{6}}}} \right)^{{\text{ + 2}}}}$?
Answer
587.1k+ views
Hint For finding the particular charge of ammonia (${\text{N}}{{\text{H}}_{\text{3}}}$), first we have to calculate the formal charge of whole molecule by adding the formal charge of each atom present in the ammonia.
Complete step by step solution:
As we know that structure of ammonia is shown as-
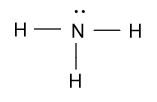
And formal charge of any compound will be calculated as follow:
Formal Charge (F.C. ) = No. of valence electrons – No. of non - bonded electrons – No. of bonds
First we calculate formal charge of each atom present in the ammonia molecule.
-F.C. on Nitrogen (${\text{N}}$) = $5 - 2 - 3 = 0$
-F.C. on each hydrogen atom (${\text{H}}$) = $1 - 0 - 1 = 0$
-From above two data it is clear that formal charge on ammonia molecules is zero and it is a neutral molecule.
Now we are known about the charge of particular compound like ammonia by which we calculate the oxidation state of given compound i.e. ${\left( {{\text{Ni}}{{\left( {{\text{N}}{{\text{H}}_{\text{3}}}} \right)}_{\text{6}}}} \right)^{{\text{ + 2}}}}$. And oxidation state of metal Nickel $\left( {{\text{Ni}}} \right)$ is calculated as follow:
-Let us take the oxidation state of nickel is $x$, and charge of ammonia is zero, then we get
$x - 0 = + 2$
Or $x = + 2$
Hence oxidation state of metal Nickel in the compound ${\left( {{\text{Ni}}{{\left( {{\text{N}}{{\text{H}}_{\text{3}}}} \right)}_{\text{6}}}} \right)^{{\text{ + 2}}}}$ is $ + 2$.
Note: In this question some of you may do wrong calculation if you are not familiar with the atomic number as well as with the electronic configuration of the atoms present in the ammonia molecule.
Complete step by step solution:
As we know that structure of ammonia is shown as-

And formal charge of any compound will be calculated as follow:
Formal Charge (F.C. ) = No. of valence electrons – No. of non - bonded electrons – No. of bonds
First we calculate formal charge of each atom present in the ammonia molecule.
-F.C. on Nitrogen (${\text{N}}$) = $5 - 2 - 3 = 0$
-F.C. on each hydrogen atom (${\text{H}}$) = $1 - 0 - 1 = 0$
-From above two data it is clear that formal charge on ammonia molecules is zero and it is a neutral molecule.
Now we are known about the charge of particular compound like ammonia by which we calculate the oxidation state of given compound i.e. ${\left( {{\text{Ni}}{{\left( {{\text{N}}{{\text{H}}_{\text{3}}}} \right)}_{\text{6}}}} \right)^{{\text{ + 2}}}}$. And oxidation state of metal Nickel $\left( {{\text{Ni}}} \right)$ is calculated as follow:
-Let us take the oxidation state of nickel is $x$, and charge of ammonia is zero, then we get
$x - 0 = + 2$
Or $x = + 2$
Hence oxidation state of metal Nickel in the compound ${\left( {{\text{Ni}}{{\left( {{\text{N}}{{\text{H}}_{\text{3}}}} \right)}_{\text{6}}}} \right)^{{\text{ + 2}}}}$ is $ + 2$.
Note: In this question some of you may do wrong calculation if you are not familiar with the atomic number as well as with the electronic configuration of the atoms present in the ammonia molecule.
Recently Updated Pages
Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Class 11 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 Biology: Engaging Questions & Answers for Success

Master Class 11 Physics: Engaging Questions & Answers for Success

Master Class 11 Accountancy: Engaging Questions & Answers for Success

Trending doubts
There are 720 permutations of the digits 1 2 3 4 5 class 11 maths CBSE

In what year Guru Nanak Dev ji was born A15 April 1469 class 11 social science CBSE

1 Quintal is equal to a 110 kg b 10 kg c 100kg d 1000 class 11 physics CBSE

10 examples of friction in our daily life

Explain zero factorial class 11 maths CBSE

When guru nanak ji was born in 1469 AD according to class 11 social studies CBSE




