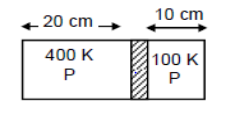
Figure shows a cylindrical tube of length 30 cm which is partitioned by a tight-fitting separator. The separator is very weakly conducting and can freely slide along the tube. Ideal gases are filled in the two parts of the vessel. In the beginning, the temperature in the parts A and B are 400 K and 100 K respectively. The separator slides to a momentary equilibrium position shown in figure. Find the final equilibrium position of the separator, reached after a long time.

Answer
601.5k+ views
Hint: Since, temperatures in the parts A and B are given. At first equilibrium, both side pressures will be the same, and putting combined gas equation of one side of the separating wall,
\[\dfrac{{{P_{{A_1}}} \times {V_{{A_1}}}}}{{{T_{{A_1}}}}} = \dfrac{{{P_{{A_2}}} \times {V_{{A_2}}}}}{{{T_{{A_2}}}}}\] and
at second equilibrium, pressures on both sides will be the same.
Complete step by step answer:
Let the initial pressure of the chambers A and B be \[{P_{{A_1}}}\] and \[{P_{{B_1}}}\], respectively.
Let the final pressure of chambers A and B be \[{P_{{A_2}}}\] and \[{P_{{B_2}}}\], respectively.
Let the Curved Surface Area be A.
\[
{V_{{A_1}}} = 0.2A \\
{T_{{A_1}}} = 400K \\
{V_{{B_1}}} = 0.1A \\
{T_{{B_1}}} = 100K \\
\]
At first equilibrium, both side pressures will be the same.
\[{P_{{A_1}}} = {P_{{B_1}}}\]
Let the final temperature at equilibrium be T. Then,
Putting combined gas equation of one side of the separating wall,
\[
\dfrac{{{P_{{A_1}}} \times {V_{{A_1}}}}}{{{T_{{A_1}}}}} = \dfrac{{{P_{{A_2}}} \times {V_{{A_2}}}}}{T} \\
\Rightarrow \dfrac{{{P_{{A_1}}} \times 0.2A}}{{400}} = \dfrac{{{P_{{A_2}}} \times {V_{{A_2}}}}}{T} \\
\]
\[ \Rightarrow {P_{{A_2}}} = \dfrac{{{P_{{A_1}}} \times T \times 0.2A}}{{400 \times {V_{{A_2}}}}}\] … (1)
For second chamber:
\[
\dfrac{{{P_{{B_1}}} \times {V_{{B_1}}}}}{{{T_{{B_1}}}}} = \dfrac{{{P_{{B_2}}} \times {V_{{B_2}}}}}{T} \\
\Rightarrow \dfrac{{{P_{{B_1}}} \times 0.1A}}{{100}} = \dfrac{{{P_{{B_2}}} \times {V_{{B_2}}}}}{T} \\
\]
\[ \Rightarrow {P_{{B_2}}} = \dfrac{{{P_{{B_1}}} \times T \times 0.1A}}{{100 \times {V_{{B_2}}}}}\] … (2)
At second equilibrium, pressures on both sides will be the same
\[
{P_{{A_2}}} = {P_{{B_2}}} \\
\Rightarrow \dfrac{{{P_{{A_1}}} \times T \times 0.2A}}{{400 \times {V_{{A_2}}}}} = \dfrac{{{P_{{B_1}}} \times T \times 0.1A}}{{100 \times {V_{{B_2}}}}} \\
\Rightarrow \dfrac{{{P_{{A_1}}}}}{{2{V_{{A_2}}}}} = \dfrac{{{P_{{B_1}}}}}{{{V_{{B_2}}}}} \\
\]
\[ \Rightarrow 2{V_{{A_2}}} = {V_{{B_2}}}\] … (3)
Now,
\[{V_{{B_2}}} + {V_{{A_2}}} = 0.3A\]
\[ \Rightarrow 2{V_{{A_2}}} + {V_{{A_2}}} = 0.3A\] (from equation (3))
$
\Rightarrow 3{V_{{A_2}}} = 0.3A \\
\Rightarrow {V_{{A_2}}} = 0.1A \\
$
Let \[{V_{{A_2}}}\] be l A
l = 0.1A
l = 10 CM
Thus, the separator will be at a distance 10 cm from the left end.
Note: The alternate way of solving this problem is,
The final position of the separating wall is at distance x from the left end. So it is at a distance \[(30-x)\] from the right end.
Putting combined gas equation of one side of the separating wall,
$\dfrac{{{P_1} \times {V_1}}}{{{T_1}}} = \dfrac{{{P_2} \times {V_2}}}{{{T_2}}}$
$ \Rightarrow \dfrac{{{P_1} \times 20A}}{{400}} = \dfrac{{{P_2} \times A}}{T}$ … (a)
$ \Rightarrow \dfrac{{{P_1} \times 10A}}{{100}} = \dfrac{{ - {P_2} \times \left( {30 - x} \right)}}{T}$ … (b)
From equation (a) and (b), we get
$
\dfrac{1}{2} = \dfrac{x}{{30 - x}} \\
\Rightarrow 30 - x = 2x \\
\Rightarrow 3x = 30 \\
\Rightarrow x = 10cm \\
$
\[\dfrac{{{P_{{A_1}}} \times {V_{{A_1}}}}}{{{T_{{A_1}}}}} = \dfrac{{{P_{{A_2}}} \times {V_{{A_2}}}}}{{{T_{{A_2}}}}}\] and
at second equilibrium, pressures on both sides will be the same.
Complete step by step answer:
Let the initial pressure of the chambers A and B be \[{P_{{A_1}}}\] and \[{P_{{B_1}}}\], respectively.
Let the final pressure of chambers A and B be \[{P_{{A_2}}}\] and \[{P_{{B_2}}}\], respectively.
Let the Curved Surface Area be A.
\[
{V_{{A_1}}} = 0.2A \\
{T_{{A_1}}} = 400K \\
{V_{{B_1}}} = 0.1A \\
{T_{{B_1}}} = 100K \\
\]
At first equilibrium, both side pressures will be the same.
\[{P_{{A_1}}} = {P_{{B_1}}}\]
Let the final temperature at equilibrium be T. Then,
Putting combined gas equation of one side of the separating wall,
\[
\dfrac{{{P_{{A_1}}} \times {V_{{A_1}}}}}{{{T_{{A_1}}}}} = \dfrac{{{P_{{A_2}}} \times {V_{{A_2}}}}}{T} \\
\Rightarrow \dfrac{{{P_{{A_1}}} \times 0.2A}}{{400}} = \dfrac{{{P_{{A_2}}} \times {V_{{A_2}}}}}{T} \\
\]
\[ \Rightarrow {P_{{A_2}}} = \dfrac{{{P_{{A_1}}} \times T \times 0.2A}}{{400 \times {V_{{A_2}}}}}\] … (1)
For second chamber:
\[
\dfrac{{{P_{{B_1}}} \times {V_{{B_1}}}}}{{{T_{{B_1}}}}} = \dfrac{{{P_{{B_2}}} \times {V_{{B_2}}}}}{T} \\
\Rightarrow \dfrac{{{P_{{B_1}}} \times 0.1A}}{{100}} = \dfrac{{{P_{{B_2}}} \times {V_{{B_2}}}}}{T} \\
\]
\[ \Rightarrow {P_{{B_2}}} = \dfrac{{{P_{{B_1}}} \times T \times 0.1A}}{{100 \times {V_{{B_2}}}}}\] … (2)
At second equilibrium, pressures on both sides will be the same
\[
{P_{{A_2}}} = {P_{{B_2}}} \\
\Rightarrow \dfrac{{{P_{{A_1}}} \times T \times 0.2A}}{{400 \times {V_{{A_2}}}}} = \dfrac{{{P_{{B_1}}} \times T \times 0.1A}}{{100 \times {V_{{B_2}}}}} \\
\Rightarrow \dfrac{{{P_{{A_1}}}}}{{2{V_{{A_2}}}}} = \dfrac{{{P_{{B_1}}}}}{{{V_{{B_2}}}}} \\
\]
\[ \Rightarrow 2{V_{{A_2}}} = {V_{{B_2}}}\] … (3)
Now,
\[{V_{{B_2}}} + {V_{{A_2}}} = 0.3A\]
\[ \Rightarrow 2{V_{{A_2}}} + {V_{{A_2}}} = 0.3A\] (from equation (3))
$
\Rightarrow 3{V_{{A_2}}} = 0.3A \\
\Rightarrow {V_{{A_2}}} = 0.1A \\
$
Let \[{V_{{A_2}}}\] be l A
l = 0.1A
l = 10 CM
Thus, the separator will be at a distance 10 cm from the left end.
Note: The alternate way of solving this problem is,
The final position of the separating wall is at distance x from the left end. So it is at a distance \[(30-x)\] from the right end.
Putting combined gas equation of one side of the separating wall,
$\dfrac{{{P_1} \times {V_1}}}{{{T_1}}} = \dfrac{{{P_2} \times {V_2}}}{{{T_2}}}$
$ \Rightarrow \dfrac{{{P_1} \times 20A}}{{400}} = \dfrac{{{P_2} \times A}}{T}$ … (a)
$ \Rightarrow \dfrac{{{P_1} \times 10A}}{{100}} = \dfrac{{ - {P_2} \times \left( {30 - x} \right)}}{T}$ … (b)
From equation (a) and (b), we get
$
\dfrac{1}{2} = \dfrac{x}{{30 - x}} \\
\Rightarrow 30 - x = 2x \\
\Rightarrow 3x = 30 \\
\Rightarrow x = 10cm \\
$
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Trending doubts
Which is more stable and why class 12 chemistry CBSE

Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

What are the major means of transport Explain each class 12 social science CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE




