Explain what is meant by the pyranose structure of glucose.
Answer
600.9k+ views
Hint:The pyranose is the chemical structure which contains six membered rings where five carbon atoms are present and one oxygen atom is present. The pyranose structures are seen in saccharides.
Complete step by step answer:The pyranose ring of glucose is formed by reacting the hydroxyl group present in the fifth position of carbon (C-5) of the glucose molecule with the aldehyde group attached to the first position of the carbon (C-1) of the same glucose molecule. This results in the formation of intramolecular hemiacetal molecules.
The name pyranose is taken from the heterocyclic compound containing an oxygen atom with five carbon atoms forming a cyclic structure known as pyran.
The structure of the pyranose ring of glucose is shown below.
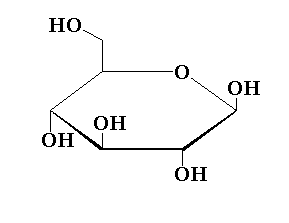
In this pyranose ring structure of glucose, five carbon atoms are present and one oxygen atom is present forming a six membered ring structure.
There are a total 38 conformation structures of pyranose ring, 2 chair form, 6 boats form, 6 skew boat form, 12 half-chairs form and 12 envelopes forms.
Note:When the hydroxyl group present on the fourth position of the carbon (C-4) of the glucose molecule reacts with the aldehydic group present in the first position of carbon (C-1) of the same glucose molecule then furanose is formed. In furanose, four carbon atoms and one oxygen atom are present forming a five membered cyclic furanose ring. The pyranose ring is thermally more stable than the furanose ring.
Complete step by step answer:The pyranose ring of glucose is formed by reacting the hydroxyl group present in the fifth position of carbon (C-5) of the glucose molecule with the aldehyde group attached to the first position of the carbon (C-1) of the same glucose molecule. This results in the formation of intramolecular hemiacetal molecules.
The name pyranose is taken from the heterocyclic compound containing an oxygen atom with five carbon atoms forming a cyclic structure known as pyran.
The structure of the pyranose ring of glucose is shown below.

In this pyranose ring structure of glucose, five carbon atoms are present and one oxygen atom is present forming a six membered ring structure.
There are a total 38 conformation structures of pyranose ring, 2 chair form, 6 boats form, 6 skew boat form, 12 half-chairs form and 12 envelopes forms.
Note:When the hydroxyl group present on the fourth position of the carbon (C-4) of the glucose molecule reacts with the aldehydic group present in the first position of carbon (C-1) of the same glucose molecule then furanose is formed. In furanose, four carbon atoms and one oxygen atom are present forming a five membered cyclic furanose ring. The pyranose ring is thermally more stable than the furanose ring.
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 Chemistry: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Name the crygenes that control cotton bollworm and class 12 biology CBSE

Ribosomal RNA is actively synthesised in A Nucleoplasm class 12 biology CBSE

How many molecules of ATP and NADPH are required information class 12 biology CBSE

In a transcription unit the promoter is said to be class 12 biology CBSE

The number of chromosomes in male grasshopper is a class 12 biology CBSE




