Explain the mechanism of \[{S_N}2\] reaction with one example.
Answer
615.9k+ views
Hint: \[{S_N}2\] reaction mechanism requires the attack of nucleophiles from the back side of the carbon atom, resulting in a stereochemical position opposite to the leaving group. It is an example of stereospecific reaction.
Complete step-by-step solution:
The \[{S_N}2\] reaction is a nucleophilic substitution reaction where one bond is broken, and another is formed simultaneously. The two reacting species that are involved in the rate determining step of the reaction. Substitution nucleophilic by molecular reaction is also referred to as associative substitution and interchange mechanism.
This reaction is a second-order reaction, in which the rate determining step depends on concentration of nucleophile and the substrate. The reaction proceeds through a backside attack of nucleophiles on the substrate at an angle of $180^0$ to the carbon-leaving bond. The carbon-nucleophile bond forms and carbon-leaving bond breaks simultaneously via a transition state involved.
Now, the leaving group is pushed out of the transition state forming the required product on the opposite side of the carbon-nucleophile bond. The product formed is an inversion of the tetrahedral geometry at the central atom.
There are two ways in which a nucleophile can attack the substrate. A front side attack results in retention of stereochemical configuration in the product. A backside attack results in the inversion of stereochemical configuration in the product. As we get a hundred percent inversion in stereochemical configuration of \[{S_N}2\] reactions, it occurs through a backside attack.
For example,
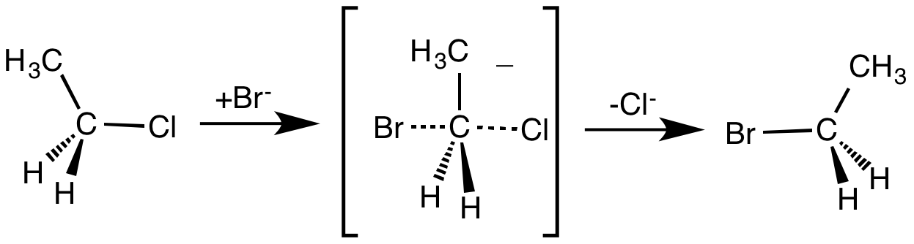
Note: Primary substrates undergo nucleophilic substitution easily as their back is unhindered. Nucleophilicity increases with a more negative charge and speeds up the rate of reaction. Polar aprotic solvents do not hinder the nucleophile and acetone is one such good solvent.
Complete step-by-step solution:
The \[{S_N}2\] reaction is a nucleophilic substitution reaction where one bond is broken, and another is formed simultaneously. The two reacting species that are involved in the rate determining step of the reaction. Substitution nucleophilic by molecular reaction is also referred to as associative substitution and interchange mechanism.
This reaction is a second-order reaction, in which the rate determining step depends on concentration of nucleophile and the substrate. The reaction proceeds through a backside attack of nucleophiles on the substrate at an angle of $180^0$ to the carbon-leaving bond. The carbon-nucleophile bond forms and carbon-leaving bond breaks simultaneously via a transition state involved.
Now, the leaving group is pushed out of the transition state forming the required product on the opposite side of the carbon-nucleophile bond. The product formed is an inversion of the tetrahedral geometry at the central atom.
There are two ways in which a nucleophile can attack the substrate. A front side attack results in retention of stereochemical configuration in the product. A backside attack results in the inversion of stereochemical configuration in the product. As we get a hundred percent inversion in stereochemical configuration of \[{S_N}2\] reactions, it occurs through a backside attack.
For example,

Note: Primary substrates undergo nucleophilic substitution easily as their back is unhindered. Nucleophilicity increases with a more negative charge and speeds up the rate of reaction. Polar aprotic solvents do not hinder the nucleophile and acetone is one such good solvent.
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Trending doubts
Which is more stable and why class 12 chemistry CBSE

Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

What are the major means of transport Explain each class 12 social science CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE




