EDTA has coordination number
(A) 3
(B) 4
(C) 5
(D) 6
Answer
532.5k+ views
Hint :The total number of atoms, ions, or molecules bound to an atom in a particular molecule or crystal is referred to as the coordination number of that atom. The coordination number of an atom is sometimes referred to as its ligancy. The ligands are the atoms, ions, or molecules that are bound to the centre atom (or molecule/ion). When determining the coordination number of a central atom in a crystal, the ligancy of molecules is computed differently.
Complete Step By Step Answer:
The aminopolycarboxylic acid ethylenediaminetetraacetic acid (EDTA) has the formula $ {[C{H_2}N{(C{H_2}C{O_2}H)_2}]_2} $ . This water-soluble white substance is commonly used to bind iron and calcium ions. As a hexadentate ("six-toothed") chelating agent, it binds these ions. EDTA comes in a variety of forms, including disodium EDTA, sodium calcium edetate, and tetrasodium EDTA. $ EDT{A^{4 - }} $ belongs to the aminopolycarboxylic acid family of ligands in coordination chemistry. The two amines and four carboxylates in $ EDT{A^{4 - }} $ typically bond to a metal cation.
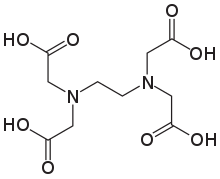
The coordination number for EDTA is 6. The coordinate number of the metal or ion is the number of ligand atoms that are directly attached to the core metal atom or ion by the coordinate bond. It's the number of chemical bonds formed between the ligand and the core metal atom or ion.
Hence option d is correct.
Note :
Concerns regarding the biodegradability of aminopolycarboxylates like EDTA have sparked interest in environmental safety. Alternative aminopolycarboxylates are being investigated as a result of these issues. Nitrilotriacetic acid (NTA), iminodisuccinic acid (IDS), polyaspartic acid, S,S-ethylenediamine-N,N′-disuccinic acid (EDDS), methylglycinediacetic acid (MGDA), and L-Glutamic acid N,N-diacetic acid, tetrasodium salt are examples of potential chelating agents (GLDA).
Complete Step By Step Answer:
The aminopolycarboxylic acid ethylenediaminetetraacetic acid (EDTA) has the formula $ {[C{H_2}N{(C{H_2}C{O_2}H)_2}]_2} $ . This water-soluble white substance is commonly used to bind iron and calcium ions. As a hexadentate ("six-toothed") chelating agent, it binds these ions. EDTA comes in a variety of forms, including disodium EDTA, sodium calcium edetate, and tetrasodium EDTA. $ EDT{A^{4 - }} $ belongs to the aminopolycarboxylic acid family of ligands in coordination chemistry. The two amines and four carboxylates in $ EDT{A^{4 - }} $ typically bond to a metal cation.

The coordination number for EDTA is 6. The coordinate number of the metal or ion is the number of ligand atoms that are directly attached to the core metal atom or ion by the coordinate bond. It's the number of chemical bonds formed between the ligand and the core metal atom or ion.
Hence option d is correct.
Note :
Concerns regarding the biodegradability of aminopolycarboxylates like EDTA have sparked interest in environmental safety. Alternative aminopolycarboxylates are being investigated as a result of these issues. Nitrilotriacetic acid (NTA), iminodisuccinic acid (IDS), polyaspartic acid, S,S-ethylenediamine-N,N′-disuccinic acid (EDDS), methylglycinediacetic acid (MGDA), and L-Glutamic acid N,N-diacetic acid, tetrasodium salt are examples of potential chelating agents (GLDA).
Recently Updated Pages
Master Class 9 General Knowledge: Engaging Questions & Answers for Success

Master Class 9 Maths: Engaging Questions & Answers for Success

Master Class 9 Science: Engaging Questions & Answers for Success

Master Class 9 English: Engaging Questions & Answers for Success

Master Class 9 Social Science: Engaging Questions & Answers for Success

Class 9 Question and Answer - Your Ultimate Solutions Guide

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE

Why is the cell called the structural and functional class 12 biology CBSE

A dentist uses a small mirror that gives a magnification class 12 physics CBSE




