Draw the structure of the monomer for each of the following polymers:
1. Nylon 6
2. Polypropene
Answer
631.5k+ views
Hint: We know that these two are polymers. We have to draw the structures of the single unit, so that by repeating those units we can get these polymers.
Step by step solution:
We know that a monomer is a molecule that can be bonded to other identical molecules to form a polymer.
(1) Nylon 6: It is also known as polycaprolactam. It is a polymer formed by ring – opening polymerization of caprolactam. So, the polymer of Nylon 6 is g-caprolactam. During polymerization, the amide bond within each caprolactam molecule is broken. Here is the structure:
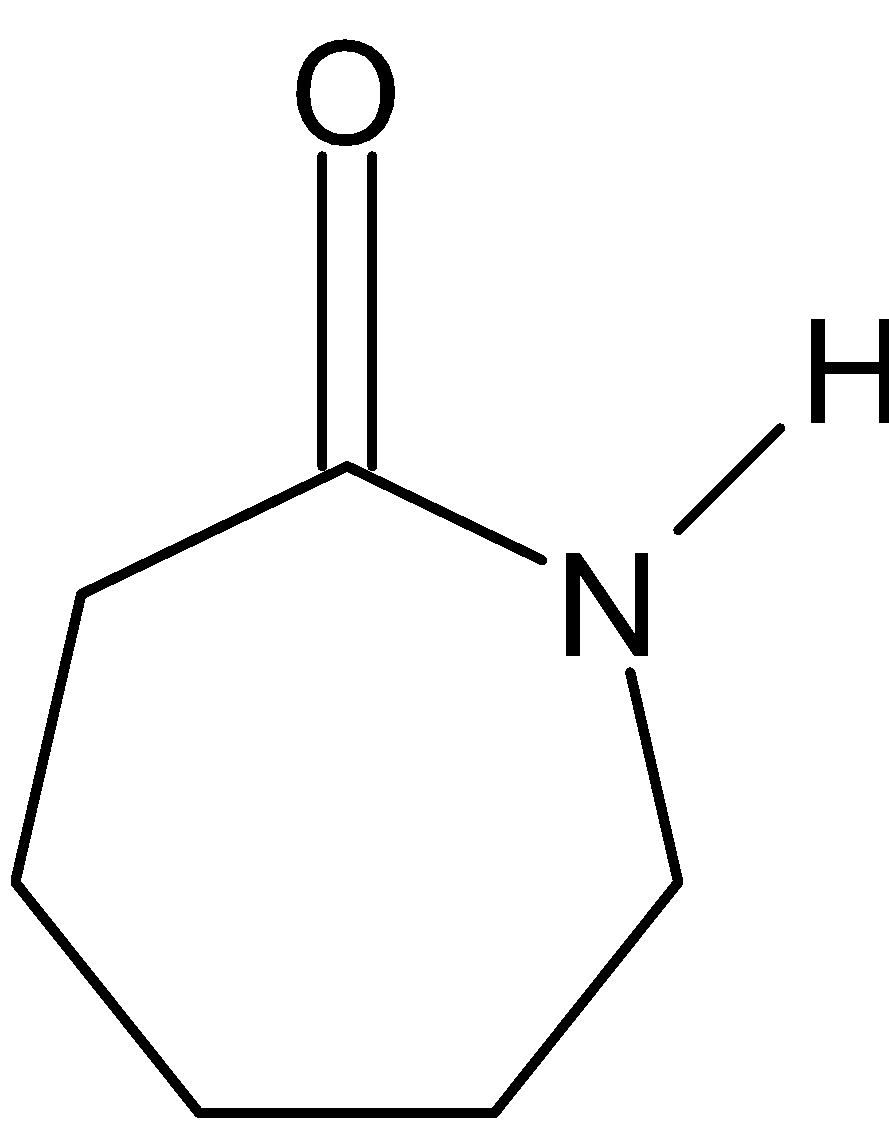
(2) Polypropene: It is also known as polypropene. It is a thermoplastic polymer used in a wide variety of applications. It is produced via chain-growth polymerization from the monomer propylene. It is made by addition polymerization. It is used in a variety of applications to include packaging for consumer products, plastic parts for various industries including the automotive industry, special devices like living hinges, and textiles. Here is the structure of monomer:
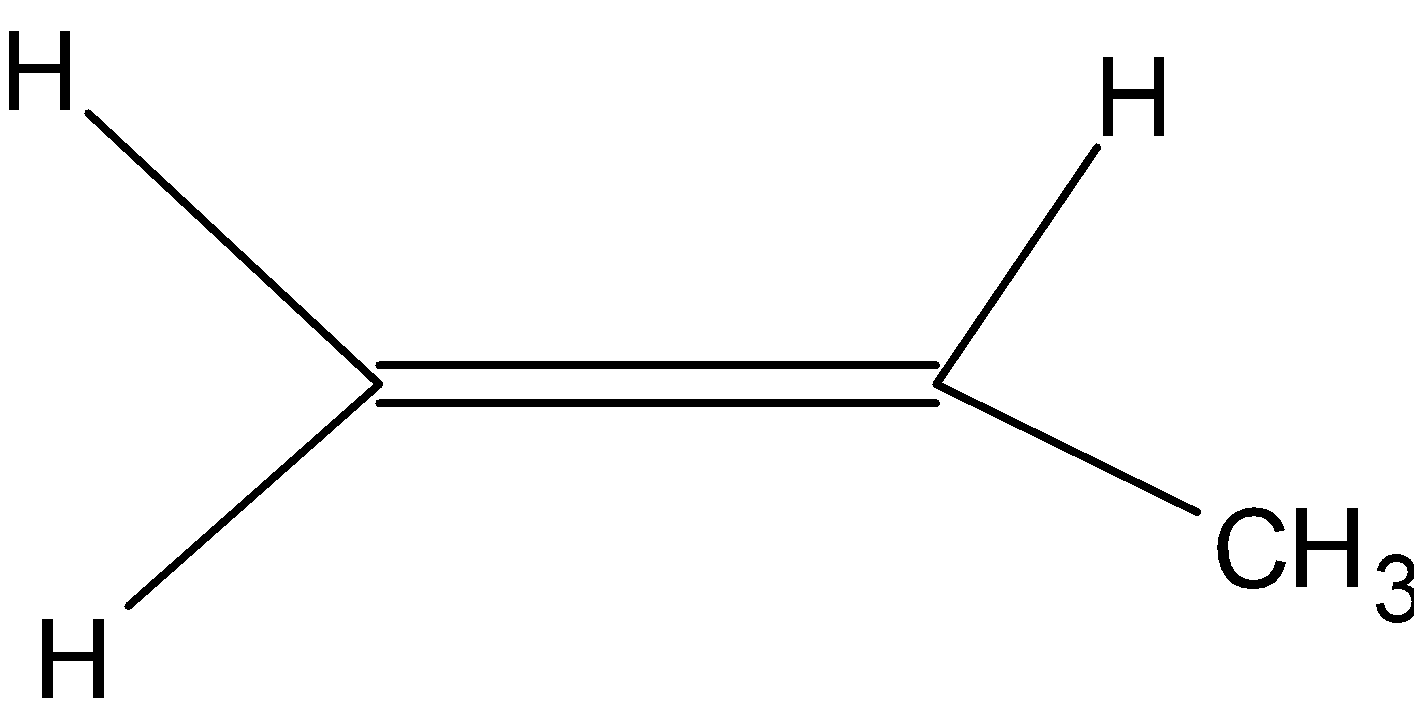
Note: Nylon 6 is not a condensation polymer. Polypropylene belongs to the group of polyolefins and is partially crystalline and non-polar. Polypropylene (PP) is a thermoplastic.
Step by step solution:
We know that a monomer is a molecule that can be bonded to other identical molecules to form a polymer.
(1) Nylon 6: It is also known as polycaprolactam. It is a polymer formed by ring – opening polymerization of caprolactam. So, the polymer of Nylon 6 is g-caprolactam. During polymerization, the amide bond within each caprolactam molecule is broken. Here is the structure:

(2) Polypropene: It is also known as polypropene. It is a thermoplastic polymer used in a wide variety of applications. It is produced via chain-growth polymerization from the monomer propylene. It is made by addition polymerization. It is used in a variety of applications to include packaging for consumer products, plastic parts for various industries including the automotive industry, special devices like living hinges, and textiles. Here is the structure of monomer:

Note: Nylon 6 is not a condensation polymer. Polypropylene belongs to the group of polyolefins and is partially crystalline and non-polar. Polypropylene (PP) is a thermoplastic.
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

Why is the cell called the structural and functional class 12 biology CBSE

Draw ray diagrams each showing i myopic eye and ii class 12 physics CBSE

Which is the correct genotypic ratio of mendel dihybrid class 12 biology CBSE




