Draw the molecular orbital diagram of ${N_2}$ and calculate the bond order.
Answer
533.1k+ views
Hint: The molecular orbital theory is primarily used for explaining the type of bonding that exists in a molecule which cannot be explained by valence bond theory. The theory explains the geometry and bonding of the molecule which involve some form of resonance.
Complete answer: The basic rules of molecular orbital theory are as follows:
First principle: The number of molecular orbitals formed is always equal to the total number of atomic orbitals that have combined to form bonds.
Second principle: Bonding molecular orbitals always comprises lower energy as compared to antibonding molecular orbitals.
Third principle: Electrons of the molecule are always assigned to the orbitals in the increasing order of the orbital energy.
Fourth principle: atomic orbitals of similar energy combine to form most effective molecular orbitals.
The molecular orbital diagram of ${N_2}$ is shown as follows:
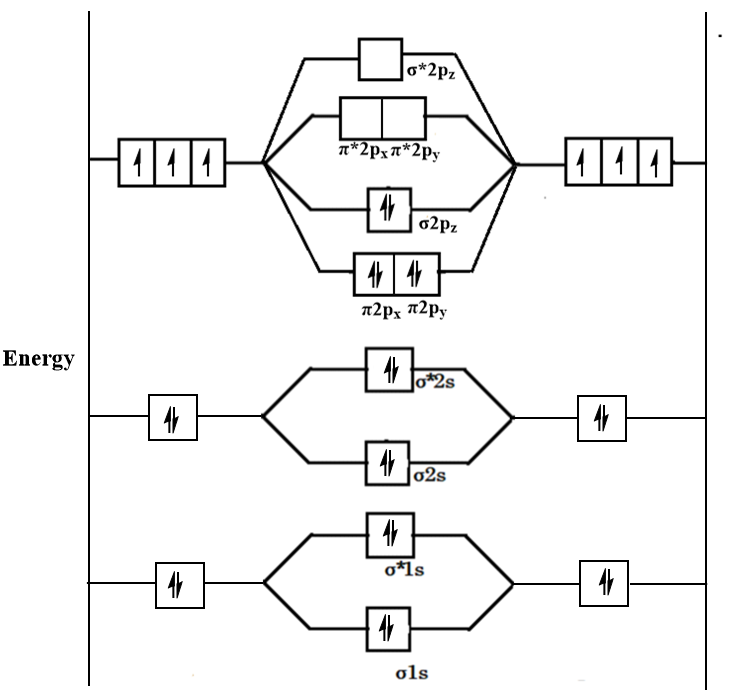
Now, the bond order of a molecule is determined by the formula which is as follows:
$BO = \dfrac{{BMO - ABMO}}{2}$
Where, BMO represents the number of electrons present in the bonding molecular orbital and ABMO represents the number of electrons in antibonding molecular orbital.
For the given molecular orbital diagram, the value of BMO is 10 and the value of ABMO is 4. Therefore, the bond order will be as follows:
$BO = \dfrac{{10 - 4}}{2} \Rightarrow 3$
Thus, we can conclude that the bond order of ${N_2}$ is 3.
Note:
It is important to note that all the elements in the second period before oxygen consist of small energy difference between 2s and 2p orbitals so that he s-p mixing can occur lowering the energy of $\sigma 2s$ and ${\sigma ^*}2s$ and increase the energy of $\sigma 2p$ and ${\sigma ^*}2p$. But on moving right in a period, the s-orbital becomes more stabilized than p-orbital and the difference in their energies increases due to which the s-p mixing of oxygen becomes smaller.
Complete answer: The basic rules of molecular orbital theory are as follows:
First principle: The number of molecular orbitals formed is always equal to the total number of atomic orbitals that have combined to form bonds.
Second principle: Bonding molecular orbitals always comprises lower energy as compared to antibonding molecular orbitals.
Third principle: Electrons of the molecule are always assigned to the orbitals in the increasing order of the orbital energy.
Fourth principle: atomic orbitals of similar energy combine to form most effective molecular orbitals.
The molecular orbital diagram of ${N_2}$ is shown as follows:

Now, the bond order of a molecule is determined by the formula which is as follows:
$BO = \dfrac{{BMO - ABMO}}{2}$
Where, BMO represents the number of electrons present in the bonding molecular orbital and ABMO represents the number of electrons in antibonding molecular orbital.
For the given molecular orbital diagram, the value of BMO is 10 and the value of ABMO is 4. Therefore, the bond order will be as follows:
$BO = \dfrac{{10 - 4}}{2} \Rightarrow 3$
Thus, we can conclude that the bond order of ${N_2}$ is 3.
Note:
It is important to note that all the elements in the second period before oxygen consist of small energy difference between 2s and 2p orbitals so that he s-p mixing can occur lowering the energy of $\sigma 2s$ and ${\sigma ^*}2s$ and increase the energy of $\sigma 2p$ and ${\sigma ^*}2p$. But on moving right in a period, the s-orbital becomes more stabilized than p-orbital and the difference in their energies increases due to which the s-p mixing of oxygen becomes smaller.
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE

Why is the cell called the structural and functional class 12 biology CBSE

Which is the correct genotypic ratio of mendel dihybrid class 12 biology CBSE




