
Draw Lewis structure for the following molecules.
i) $S{F_6}$
ii) ${C_2}{H_4}$
iii) $N{O_2}$
Answer
583.2k+ views
Hint: Lewis structure was given by an American chemist Gilbert Newton Lewis. It is used to represent the structure of atoms and molecules indicating the pair of bonded and non bonded electron pairs.
Complete step by step answer:
Lewis structure or electron dot structure is a representation or a diagram of an atom, molecule or a compound. In these structures the bonds between the atoms and molecules are indicated with straight lines. The electrons on each atom if non- bonded are indicated as lone pairs.
Let us understand the steps for drawing the Lewis structures of the given molecules.
a. The total number of valence electrons has to be added around each atom in a molecule.
b. For anions extra electrons are added to the total number of electrons.
c. For cations extra electrons are subtracted from the total number of electrons.
d. The less electronegative atom is positioned at the centre and surrounded by other atoms.
e. The shared electrons or bonded electrons are shown as single bonds
f. Assign the number of lone pairs first to more electronegative than to less electronegative atoms following octet rule.
g. A double or triple bond is drawn between bonded atoms if required to satisfy the octet of each atom.
Let us draw the Lewis structure for the given molecules using the protocol.
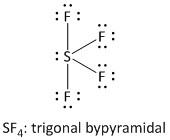
i) \[S{F_4}\] : It is a neutral molecule.
The total number of electrons = valence electrons of \[S\] atom + \[4\] x valence electrons of \[F\] atom.
= $6 + 4 \times 7 = 34$
Sulphur is the less electronegative atom and is the central atom. It has a total of five electron pairs of which four are bonded and one non-bonded lone pair. Fluorine has a total of four electron pairs of which one is bonded and three non-bonded lone pairs. The four fluorine atoms form four single bonds. Thus the structure of \[S{F_4}\] is:
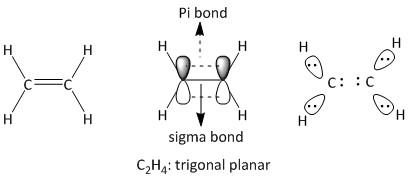
ii) \[{C_2}{H_4}\] . It is a neutral molecule known as ethene.
The total number of electrons = \[2\] x valence electrons of \[C\] atom + \[4\] x valence electrons of \[H\] atom.
= $2 \times 4 + 4 \times 1 = 12$
The electronegativity difference of carbon and hydrogen is small. There are two central carbon atoms in this molecule. Each carbon atom has a total of four electron pairs of which four are bonded. The two hydrogen atoms are bonded via two single bonds. The other two electrons on carbon are bonded to other carbon by a single and a double bond. Two sets of each hydrogen atom attached to two carbon atoms have a total of one electron pair which is bonded. Thus the structure of \[{C_2}{H_4}\] is:
iii) $N{O_2}$: It is a neutral molecule.
The total number of electrons = valence electrons of \[N\] atom + \[2\] x valence electrons of \[O\] atom
= $5 + 2 \times 6 = 17$
Nitrogen is the less electronegative atom and is the central atom. It has a total of three electron pairs which are bonded and one unpaired electron. There are two different types of oxygen. One has a total of four electron pairs of which one is bonded and three non-bonded lone pairs. The other oxygen atom has two bonded (one sigma and one double bond) and two non-bonded electron pairs. The two oxygen atoms are placed on either side of the nitrogen atom. Thus the structure of $N{O_2}$ is:

Note: Lewis assigned the compounds as bases which donates an electron pair and as acids which accept an electron pair. In some cases Lewis structure violates the octet rule like in $N{O_2}$ the nitrogen atom does not have an octet.
Complete step by step answer:
Lewis structure or electron dot structure is a representation or a diagram of an atom, molecule or a compound. In these structures the bonds between the atoms and molecules are indicated with straight lines. The electrons on each atom if non- bonded are indicated as lone pairs.
Let us understand the steps for drawing the Lewis structures of the given molecules.
a. The total number of valence electrons has to be added around each atom in a molecule.
b. For anions extra electrons are added to the total number of electrons.
c. For cations extra electrons are subtracted from the total number of electrons.
d. The less electronegative atom is positioned at the centre and surrounded by other atoms.
e. The shared electrons or bonded electrons are shown as single bonds
f. Assign the number of lone pairs first to more electronegative than to less electronegative atoms following octet rule.
g. A double or triple bond is drawn between bonded atoms if required to satisfy the octet of each atom.
Let us draw the Lewis structure for the given molecules using the protocol.
i) \[S{F_4}\] : It is a neutral molecule.
The total number of electrons = valence electrons of \[S\] atom + \[4\] x valence electrons of \[F\] atom.
= $6 + 4 \times 7 = 34$
Sulphur is the less electronegative atom and is the central atom. It has a total of five electron pairs of which four are bonded and one non-bonded lone pair. Fluorine has a total of four electron pairs of which one is bonded and three non-bonded lone pairs. The four fluorine atoms form four single bonds. Thus the structure of \[S{F_4}\] is:

ii) \[{C_2}{H_4}\] . It is a neutral molecule known as ethene.
The total number of electrons = \[2\] x valence electrons of \[C\] atom + \[4\] x valence electrons of \[H\] atom.
= $2 \times 4 + 4 \times 1 = 12$
The electronegativity difference of carbon and hydrogen is small. There are two central carbon atoms in this molecule. Each carbon atom has a total of four electron pairs of which four are bonded. The two hydrogen atoms are bonded via two single bonds. The other two electrons on carbon are bonded to other carbon by a single and a double bond. Two sets of each hydrogen atom attached to two carbon atoms have a total of one electron pair which is bonded. Thus the structure of \[{C_2}{H_4}\] is:

iii) $N{O_2}$: It is a neutral molecule.
The total number of electrons = valence electrons of \[N\] atom + \[2\] x valence electrons of \[O\] atom
= $5 + 2 \times 6 = 17$
Nitrogen is the less electronegative atom and is the central atom. It has a total of three electron pairs which are bonded and one unpaired electron. There are two different types of oxygen. One has a total of four electron pairs of which one is bonded and three non-bonded lone pairs. The other oxygen atom has two bonded (one sigma and one double bond) and two non-bonded electron pairs. The two oxygen atoms are placed on either side of the nitrogen atom. Thus the structure of $N{O_2}$ is:

Note: Lewis assigned the compounds as bases which donates an electron pair and as acids which accept an electron pair. In some cases Lewis structure violates the octet rule like in $N{O_2}$ the nitrogen atom does not have an octet.
Recently Updated Pages
Basicity of sulphurous acid and sulphuric acid are

Master Class 11 Business Studies: Engaging Questions & Answers for Success

Master Class 11 Computer Science: Engaging Questions & Answers for Success

Master Class 11 Economics: Engaging Questions & Answers for Success

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Explain zero factorial class 11 maths CBSE

State and prove Bernoullis theorem class 11 physics CBSE

What steps did the French revolutionaries take to create class 11 social science CBSE

The transition element that has lowest enthalpy of class 11 chemistry CBSE

Can anyone list 10 advantages and disadvantages of friction




