Draw energy band diagrams of an n-type and p-type semiconductor at temperature $T$ > $0$. Mark the donor and acceptor energy levels with their energies. A semiconductor has equal electron and hole concentration of $6\times {{10}^{8}}{{m}^{-3}}$. On doping with certain impurity, electron concentration increases to $9\times {{10}^{12}}{{m}^{-3}}$. Calculate the new hole concentration.
Answer
553.2k+ views
Hint: As a first step, one could recall the theory that they had learnt about the mentioned semiconductors. Then you could use the same to make the energy band diagram for both. After that recall the law of mass action and then substitute accordingly to solve the second part of the question.
Formula used:
Law of mass action,
$np={{n}_{i}}^{2}$
Complete answer:
Firstly, we are asked to draw the energy band diagrams for the n-type and p-type semiconductor at some temperature less than 0. Following are the diagrams where we have clearly marked the donor and acceptor levels, fermi level, etc.
For the n-type semiconductor the energy diagram would look like as:
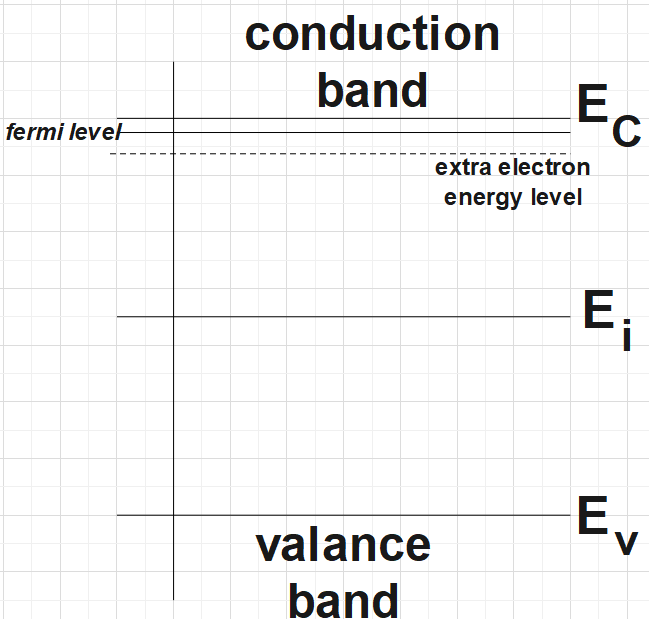
Now, for the p-type semiconductor the energy band diagram would look something like this:
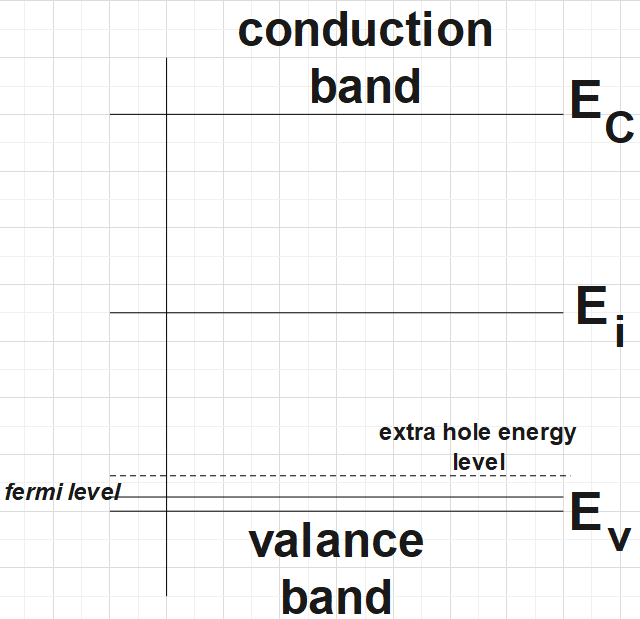
Now, let us look at the second part of the question which is a numerical problem. In order to solve this problem let us recall the law of mass action given by,
$np={{n}_{i}}^{2}$
Here we are given the intrinsic carrier concentration as,
${{n}_{i}}=6\times {{10}^{8}}{{m}^{-3}}$
Now, we are given the increased level of electron as,
$n=9\times {{10}^{12}}{{m}^{-3}}$
Now, we could substitute these values into the law of mass action to get,
$p=\dfrac{{{\left( 6\times {{10}^{8}} \right)}^{2}}}{9\times {{10}^{12}}}$
$\therefore p=4\times {{10}^{4}}{{m}^{-3}}$
Therefore, the new hole concentration to be $p=4\times {{10}^{4}}{{m}^{-3}}$.
Note:
While making the diagram one should take care that they don’t miss out any of the energy level. You could recall your basic understanding of the given semiconductors, namely, the n-type and the p-type. So, you would get the basic idea as to where acceptor level and donor level are significant.
Formula used:
Law of mass action,
$np={{n}_{i}}^{2}$
Complete answer:
Firstly, we are asked to draw the energy band diagrams for the n-type and p-type semiconductor at some temperature less than 0. Following are the diagrams where we have clearly marked the donor and acceptor levels, fermi level, etc.
For the n-type semiconductor the energy diagram would look like as:

Now, for the p-type semiconductor the energy band diagram would look something like this:

Now, let us look at the second part of the question which is a numerical problem. In order to solve this problem let us recall the law of mass action given by,
$np={{n}_{i}}^{2}$
Here we are given the intrinsic carrier concentration as,
${{n}_{i}}=6\times {{10}^{8}}{{m}^{-3}}$
Now, we are given the increased level of electron as,
$n=9\times {{10}^{12}}{{m}^{-3}}$
Now, we could substitute these values into the law of mass action to get,
$p=\dfrac{{{\left( 6\times {{10}^{8}} \right)}^{2}}}{9\times {{10}^{12}}}$
$\therefore p=4\times {{10}^{4}}{{m}^{-3}}$
Therefore, the new hole concentration to be $p=4\times {{10}^{4}}{{m}^{-3}}$.
Note:
While making the diagram one should take care that they don’t miss out any of the energy level. You could recall your basic understanding of the given semiconductors, namely, the n-type and the p-type. So, you would get the basic idea as to where acceptor level and donor level are significant.
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 Chemistry: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Name the crygenes that control cotton bollworm and class 12 biology CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

In a transcription unit the promoter is said to be class 12 biology CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE




