How would you draw constitutional isomers for the molecular formula ${{\text{C}}_{\text{4}}}{{\text{H}}_{\text{8}}}$?
Answer
582k+ views
Hint: Constitutional isomers are also known by the name of structural isomers and for drawing the constitutional structure of any compound we have to know about the number of atoms and which kind of bond is present in that molecule.
Complete step by step answer:
- Constitutional isomers are those isomers which are formed by the different arrangements of the atoms and bonds present inside the molecule.
-In the question given molecule is ${{\text{C}}_{\text{4}}}{{\text{H}}_{\text{8}}}$ and it comes under the category of alkene as it matches the general formula of alkene ${{\text{C}}_{\text{n}}}{{\text{H}}_{{\text{2n}}}}$ where the number of carbon atoms are equal to the number of hydrogen atoms.
-In the given compound all bonds are single only accepting one which is double.
-Constitutional isomers of Butene (${{\text{C}}_{\text{4}}}{{\text{H}}_{\text{8}}}$) will be formed by the different arrangement of the atoms and bonds present in it.
-In the first constitutional isomer of Butene (${{\text{C}}_{\text{4}}}{{\text{H}}_{\text{8}}}$), double bond is present at the first carbon atom whose name will be But-1-ene and it is showing as ${\text{C}}{{\text{H}}_{\text{2}}}{\text{ = CH - C}}{{\text{H}}_{\text{2}}}{\text{ - C}}{{\text{H}}_{\text{3}}}$.
-In the second constitutional isomer of Butene (${{\text{C}}_{\text{4}}}{{\text{H}}_{\text{8}}}$), double bond is present at the second carbon atom whose name will be But-2-ene and it is showing as ${\text{C}}{{\text{H}}_3}{\text{ - CH = CH - C}}{{\text{H}}_{\text{3}}}$.
-In the third constitutional isomer of Butene (${{\text{C}}_{\text{4}}}{{\text{H}}_{\text{8}}}$), substitution of methyl group is present at the second carbon atom of the propene chain and this isomer is known by the name of 2-methyl propene and it is showing as:
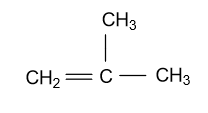
-In the fourth constitutional isomer of butene, it is present in the cyclic form whose name will be cyclo-butane and it is showing as:
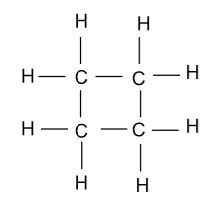
-In the fifth constitutional isomer of butene, it is present in the methyl cyclopropane form which is shown as follow:
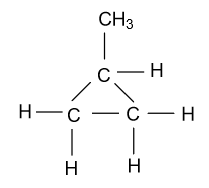
Hence by the above way we will draw the constitutional isomers for the molecular formula ${{\text{C}}_{\text{4}}}{{\text{H}}_{\text{8}}}$.
Note: Here some of you may think that how cyclic structure of butene is possible as in the butene double bond is present but in the cyclic structure only single bonds, so the reason is that in constitutional type of bond will be changed but number of atoms always remain constant.
Complete step by step answer:
- Constitutional isomers are those isomers which are formed by the different arrangements of the atoms and bonds present inside the molecule.
-In the question given molecule is ${{\text{C}}_{\text{4}}}{{\text{H}}_{\text{8}}}$ and it comes under the category of alkene as it matches the general formula of alkene ${{\text{C}}_{\text{n}}}{{\text{H}}_{{\text{2n}}}}$ where the number of carbon atoms are equal to the number of hydrogen atoms.
-In the given compound all bonds are single only accepting one which is double.
-Constitutional isomers of Butene (${{\text{C}}_{\text{4}}}{{\text{H}}_{\text{8}}}$) will be formed by the different arrangement of the atoms and bonds present in it.
-In the first constitutional isomer of Butene (${{\text{C}}_{\text{4}}}{{\text{H}}_{\text{8}}}$), double bond is present at the first carbon atom whose name will be But-1-ene and it is showing as ${\text{C}}{{\text{H}}_{\text{2}}}{\text{ = CH - C}}{{\text{H}}_{\text{2}}}{\text{ - C}}{{\text{H}}_{\text{3}}}$.
-In the second constitutional isomer of Butene (${{\text{C}}_{\text{4}}}{{\text{H}}_{\text{8}}}$), double bond is present at the second carbon atom whose name will be But-2-ene and it is showing as ${\text{C}}{{\text{H}}_3}{\text{ - CH = CH - C}}{{\text{H}}_{\text{3}}}$.
-In the third constitutional isomer of Butene (${{\text{C}}_{\text{4}}}{{\text{H}}_{\text{8}}}$), substitution of methyl group is present at the second carbon atom of the propene chain and this isomer is known by the name of 2-methyl propene and it is showing as:

-In the fourth constitutional isomer of butene, it is present in the cyclic form whose name will be cyclo-butane and it is showing as:

-In the fifth constitutional isomer of butene, it is present in the methyl cyclopropane form which is shown as follow:

Hence by the above way we will draw the constitutional isomers for the molecular formula ${{\text{C}}_{\text{4}}}{{\text{H}}_{\text{8}}}$.
Note: Here some of you may think that how cyclic structure of butene is possible as in the butene double bond is present but in the cyclic structure only single bonds, so the reason is that in constitutional type of bond will be changed but number of atoms always remain constant.
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Trending doubts
Which is more stable and why class 12 chemistry CBSE

Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

What are the major means of transport Explain each class 12 social science CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE




