Double bond equivalent of cubane is:
A.$4$
B. $5$
C. $6$
D. $7$

Answer
591k+ views
Hint:
Double bond equivalent is used to calculate the number of unsaturation present in an organic molecule. The term unsaturated means a double bond or a ring in a molecule structure. It is the total number of pi bonds present in the structure.
Formula used:
$DBE=C+1-\dfrac{H}{2}+\dfrac{N}{2}$
where, $DBE$ is the double bond equivalent, $C$ is the number of carbon atoms, $H$ is the number of hydrogen atoms, $N$ is the number of nitrogen atoms in the molecule.
Complete step by step answer:
An unsaturated molecule is a molecule that contains double bond and triple bond and can undergo additional reactions. For example, alkenes contain one double bond and have a general formula ${{C}_{n}}{{H}_{2n}}$. Double bond equivalent is used to calculate the level of unsaturation in a molecule. The formula used for double bond equivalent is:
$DBE=C+1-\dfrac{H}{2}+\dfrac{N}{2}$
where, $DBE$ is the double bond equivalent, $C$ is the number of carbon atoms, $H$ is the number of hydrogen atoms, $N$ is the number of nitrogen atoms in the molecule.
The molecular formula of cubane is ${{C}_{8}}{{H}_{8}}$. Here, the number of carbon atoms is $8$, the number of hydrogen atoms is $8$ and the number of nitrogen atoms is $0$.
On substituting the values in the above formula, we get,
$DBE=8+1-\dfrac{8}{2}+\dfrac{0}{2}$
$\Rightarrow DBE=5$
Therefore, the double bond equivalent of cubane is $5$.
Hence, the correct option is (B).
Additional information:
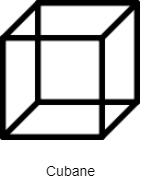
Cubane is a solid crystalline substance that has a molecular formula ${{C}_{8}}{{H}_{8}}$ and consists of eight carbon atoms that are arranged at the corners of a cube and one hydrogen atom is attached to each carbon atom. It is a synthetic hydrocarbon molecule.
Note: The structure of cubane contains six rings that correspond to the six faces of a cube.
-The molecular formula of cubane is ${{C}_{8}}{{H}_{8}}$.It is a solid crystalline substance and a member of prismanes.
-Double bond equivalent is used to determine the degree of unsaturation in a molecule.
Double bond equivalent is used to calculate the number of unsaturation present in an organic molecule. The term unsaturated means a double bond or a ring in a molecule structure. It is the total number of pi bonds present in the structure.
Formula used:
$DBE=C+1-\dfrac{H}{2}+\dfrac{N}{2}$
where, $DBE$ is the double bond equivalent, $C$ is the number of carbon atoms, $H$ is the number of hydrogen atoms, $N$ is the number of nitrogen atoms in the molecule.
Complete step by step answer:
An unsaturated molecule is a molecule that contains double bond and triple bond and can undergo additional reactions. For example, alkenes contain one double bond and have a general formula ${{C}_{n}}{{H}_{2n}}$. Double bond equivalent is used to calculate the level of unsaturation in a molecule. The formula used for double bond equivalent is:
$DBE=C+1-\dfrac{H}{2}+\dfrac{N}{2}$
where, $DBE$ is the double bond equivalent, $C$ is the number of carbon atoms, $H$ is the number of hydrogen atoms, $N$ is the number of nitrogen atoms in the molecule.
The molecular formula of cubane is ${{C}_{8}}{{H}_{8}}$. Here, the number of carbon atoms is $8$, the number of hydrogen atoms is $8$ and the number of nitrogen atoms is $0$.
On substituting the values in the above formula, we get,
$DBE=8+1-\dfrac{8}{2}+\dfrac{0}{2}$
$\Rightarrow DBE=5$
Therefore, the double bond equivalent of cubane is $5$.
Hence, the correct option is (B).
Additional information:
Cubane is a solid crystalline substance that has a molecular formula ${{C}_{8}}{{H}_{8}}$ and consists of eight carbon atoms that are arranged at the corners of a cube and one hydrogen atom is attached to each carbon atom. It is a synthetic hydrocarbon molecule.
Note: The structure of cubane contains six rings that correspond to the six faces of a cube.
-The molecular formula of cubane is ${{C}_{8}}{{H}_{8}}$.It is a solid crystalline substance and a member of prismanes.
-Double bond equivalent is used to determine the degree of unsaturation in a molecule.
Recently Updated Pages
Master Class 10 Computer Science: Engaging Questions & Answers for Success

Master Class 10 General Knowledge: Engaging Questions & Answers for Success

Master Class 10 English: Engaging Questions & Answers for Success

Master Class 10 Social Science: Engaging Questions & Answers for Success

Master Class 10 Maths: Engaging Questions & Answers for Success

Master Class 10 Science: Engaging Questions & Answers for Success

Trending doubts
Which is more stable and why class 12 chemistry CBSE

Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

What are the major means of transport Explain each class 12 social science CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE




