What does the distillation of phenol with Zinc Dust result in?
(A) ${{C}_{6}}{{H}_{6}}$
(B) ${{C}_{6}}{{H}_{5}}-{{C}_{6}}{{H}_{5}}$
(C) ${{C}_{6}}{{H}_{12}}$
(D) ${{C}_{6}}{{H}_{5}}-O-{{C}_{6}}{{H}_{5}}$
Answer
625.5k+ views
Hint: A common name for this reaction is the Baeyer reduction of aromatic oxygen-containing compounds. Here, reduction of phenol will occur.
Complete step by step solution:
Let us break this reaction down step-by-step to help facilitate better understanding of the concept.
- Let us now go through the mechanism of this reaction to understand how the formation of Benzene and Zinc Oxide (ZnO) really takes place.
- Zn shows an oxidation state of +2.
- The phenol turns into phenoxide ion and the proton that is released accepts an electron from Zn forming H radical.
- Also because of the heating there is homolytic fission of C of phenyl ring and O-
- Thus, the O-atom formed accepts an electron from Zn and forms oxide ion.
- In this way zinc metal forms zinc oxide and phenyl radical produced here forms a bond with hydrogen radical.
- However, the yield of this reaction is lower.
- The full mechanism is shown step-by-step in the following figure:
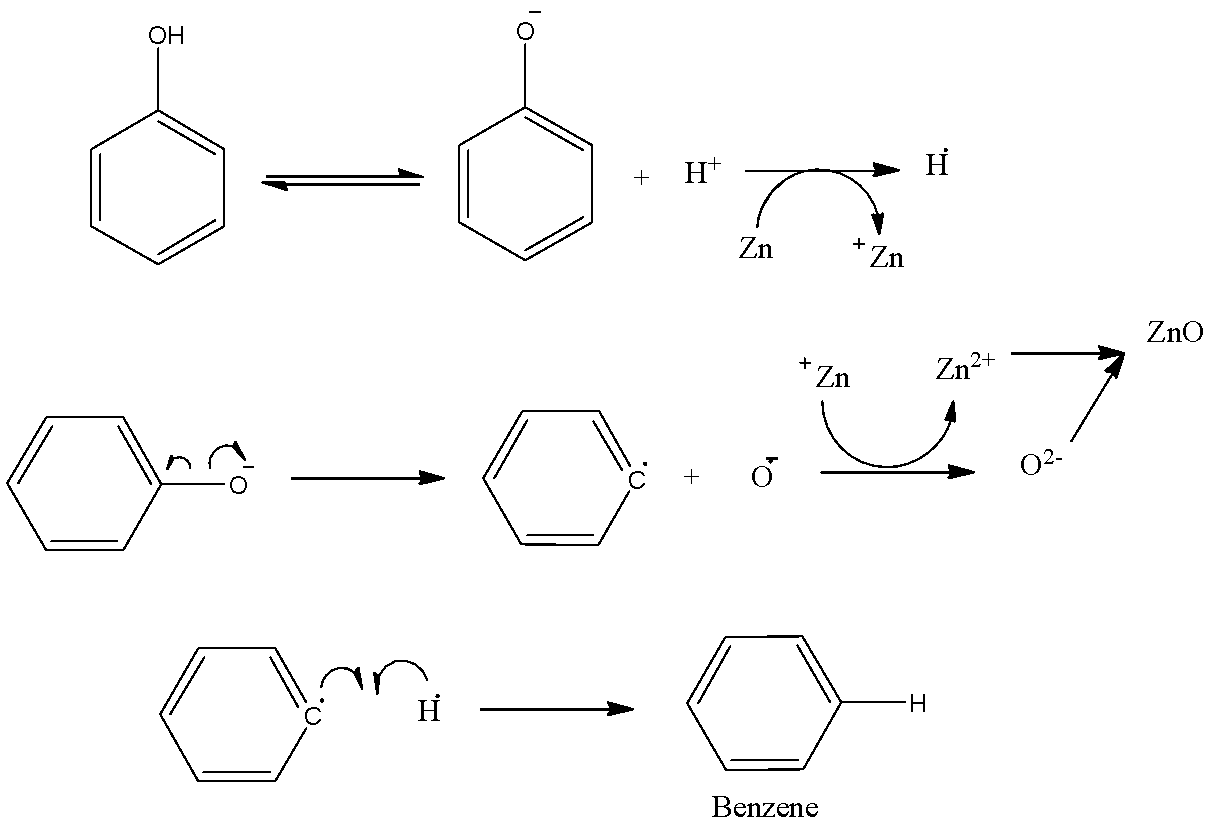
- Observe that the formation of ${{C}_{12}}{{H}_{10}}$ and ${{H}_{2}}$ is also possible due to the fusion of the benzene and the hydrogen radicals with another one of the same kinds.
- The equation of the given reaction is as follows:
\[{{C}_{6}}{{H}_{5}}-OH\xrightarrow{ZnO}{{C}_{6}}{{H}_{6}}\]
So, the correct answer is “Option A”.
Note: Do not assume that Zinc oxide will oxidise phenol as it has an oxygen atom in it. Remember that actually it is used as a reducing agent. Remember that Zinc oxide cannot reduce carbon-carbon double bonds. It can only reduce carbon-oxygen single bonds.
Complete step by step solution:
Let us break this reaction down step-by-step to help facilitate better understanding of the concept.
- Let us now go through the mechanism of this reaction to understand how the formation of Benzene and Zinc Oxide (ZnO) really takes place.
- Zn shows an oxidation state of +2.
- The phenol turns into phenoxide ion and the proton that is released accepts an electron from Zn forming H radical.
- Also because of the heating there is homolytic fission of C of phenyl ring and O-
- Thus, the O-atom formed accepts an electron from Zn and forms oxide ion.
- In this way zinc metal forms zinc oxide and phenyl radical produced here forms a bond with hydrogen radical.
- However, the yield of this reaction is lower.
- The full mechanism is shown step-by-step in the following figure:

- Observe that the formation of ${{C}_{12}}{{H}_{10}}$ and ${{H}_{2}}$ is also possible due to the fusion of the benzene and the hydrogen radicals with another one of the same kinds.
- The equation of the given reaction is as follows:
\[{{C}_{6}}{{H}_{5}}-OH\xrightarrow{ZnO}{{C}_{6}}{{H}_{6}}\]
So, the correct answer is “Option A”.
Note: Do not assume that Zinc oxide will oxidise phenol as it has an oxygen atom in it. Remember that actually it is used as a reducing agent. Remember that Zinc oxide cannot reduce carbon-carbon double bonds. It can only reduce carbon-oxygen single bonds.
Recently Updated Pages
Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 English: Engaging Questions & Answers for Success

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 12 Business Studies: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE

Why is the cell called the structural and functional class 12 biology CBSE

Draw ray diagrams each showing i myopic eye and ii class 12 physics CBSE




