What is the difference between structural isomers, geometrical isomers and enantiomers?
Answer
544.8k+ views
Hint:Isomers: The compounds that have the same molecular formula but different structural formula are known as isomers. The isomers are further divided into two types i.e., structural isomers and stereoisomers based on the type of change in the structural formula of the compound.
Complete answer:Isomers are mainly categorized into two types:
1.Structural Isomers
2.Stereoisomers
Structural isomers: The compounds having the same molecular formula but structural formula differ in the bonding of atoms present in the compound.
There are different types of structural isomers:
1.Chain isomers: Compounds having the same molecular formula but differ in the branching of carbon atoms are known as chain isomers.
Example: n-butane \[ \Rightarrow C{H_3}C{H_2}C{H_2}C{H_3}\]
Iso-butane \[ \Rightarrow C{H_3}CH(C{H_3})C{H_3}\]
In n-butane there is no branching of the chain whereas in iso-butane, a methyl group is attached to the second carbon atom. Hence these are chain isomers to each other.
2.Position isomers: Compounds having the same molecular formula but differ in the position of functional groups are known as position isomers.
Example: \[1 - \] chlorobutane \[ \Rightarrow C{H_3}C{H_2}C{H_2}C{H_2}Cl\]
\[2 - \] chlorobutane \[ \Rightarrow C{H_3}CH(Cl)C{H_2}C{H_3}\]
In \[1 - \] chlorobutane, chlorine is present at the first position whereas in \[2 - \] chlorobutane, chlorine is present at the second position in the carbon chain. Hence these are positional isomers to each other.
3.Functional isomers: Compounds having the same molecular formula but different functional groups are known as functional isomers.
Example: Propanone \[ \Rightarrow C{H_3}COC{H_3}\]
Propanal \[ \Rightarrow C{H_3}C{H_2}CHO\]
In Propanone, ketone is present as a functional group whereas in Propanal, an aldehydic group is present. Hence these are functional isomers to each other.
Stereoisomers: The compounds having the same molecular formula but differ in the arrangement of atoms in three-dimensional space are known as stereoisomers. Geometrical isomers and enantiomers are types of stereoisomers.
Geometrical isomers: It is also known as cis-trans isomer. It generally arises in the alkenes in which each unsaturated alkene consists of two different functional groups. It is further divided into two types:
1.Cis-isomer: When the same groups are present in same side of alkene then it is known as cis isomer.
Example: cis-2-butene
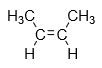
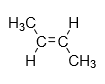
2.Trans-isomer: When the same groups are present on different side of alkene then it is known as cis isomer.
Example: trans-2-butene
Enantiomers: These are the chiral compounds which are mirror images of each other.
Example:
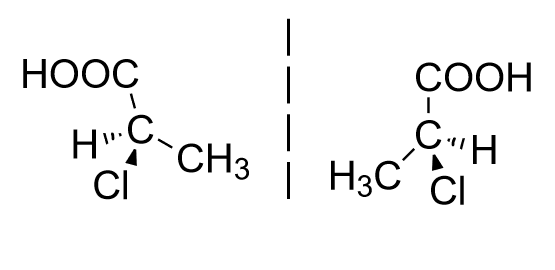
Note:
It is mandatory for a compound to be chiral to form enantiomers. Only compounds with chiral carbon can be classified as enantiomers. Enantiomers are further categorized as R-conformation and S-conformation which is done according to CIP priority rules.
Complete answer:Isomers are mainly categorized into two types:
1.Structural Isomers
2.Stereoisomers
Structural isomers: The compounds having the same molecular formula but structural formula differ in the bonding of atoms present in the compound.
There are different types of structural isomers:
1.Chain isomers: Compounds having the same molecular formula but differ in the branching of carbon atoms are known as chain isomers.
Example: n-butane \[ \Rightarrow C{H_3}C{H_2}C{H_2}C{H_3}\]
Iso-butane \[ \Rightarrow C{H_3}CH(C{H_3})C{H_3}\]
In n-butane there is no branching of the chain whereas in iso-butane, a methyl group is attached to the second carbon atom. Hence these are chain isomers to each other.
2.Position isomers: Compounds having the same molecular formula but differ in the position of functional groups are known as position isomers.
Example: \[1 - \] chlorobutane \[ \Rightarrow C{H_3}C{H_2}C{H_2}C{H_2}Cl\]
\[2 - \] chlorobutane \[ \Rightarrow C{H_3}CH(Cl)C{H_2}C{H_3}\]
In \[1 - \] chlorobutane, chlorine is present at the first position whereas in \[2 - \] chlorobutane, chlorine is present at the second position in the carbon chain. Hence these are positional isomers to each other.
3.Functional isomers: Compounds having the same molecular formula but different functional groups are known as functional isomers.
Example: Propanone \[ \Rightarrow C{H_3}COC{H_3}\]
Propanal \[ \Rightarrow C{H_3}C{H_2}CHO\]
In Propanone, ketone is present as a functional group whereas in Propanal, an aldehydic group is present. Hence these are functional isomers to each other.
Stereoisomers: The compounds having the same molecular formula but differ in the arrangement of atoms in three-dimensional space are known as stereoisomers. Geometrical isomers and enantiomers are types of stereoisomers.
Geometrical isomers: It is also known as cis-trans isomer. It generally arises in the alkenes in which each unsaturated alkene consists of two different functional groups. It is further divided into two types:
1.Cis-isomer: When the same groups are present in same side of alkene then it is known as cis isomer.
Example: cis-2-butene

2.Trans-isomer: When the same groups are present on different side of alkene then it is known as cis isomer.
Example: trans-2-butene

Enantiomers: These are the chiral compounds which are mirror images of each other.
Example:

Note:
It is mandatory for a compound to be chiral to form enantiomers. Only compounds with chiral carbon can be classified as enantiomers. Enantiomers are further categorized as R-conformation and S-conformation which is done according to CIP priority rules.
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Trending doubts
Which is more stable and why class 12 chemistry CBSE

Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

What are the major means of transport Explain each class 12 social science CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE




