Difference between homopolymer and copolymer.
Answer
572.7k+ views
Hint: To get an idea about these, separate their names into two words. Here we can write a homopolymer as homo + polymer, “homo” means the same unit and copolymer as co+polymer, where “co” means different. Now try to answer this question accordingly.
Complete step by step answer:
We should know that the main difference between copolymer and homopolymer is that copolymers are formed by using two different types of monomers, whereas homopolymers are produced by using a single type of monomer. This difference is responsible for the unique set of properties of homopolymers and copolymers.

- Polythene
- Polytetrafluoroethylene (PTFE or Teflon)
Examples of copolymers:
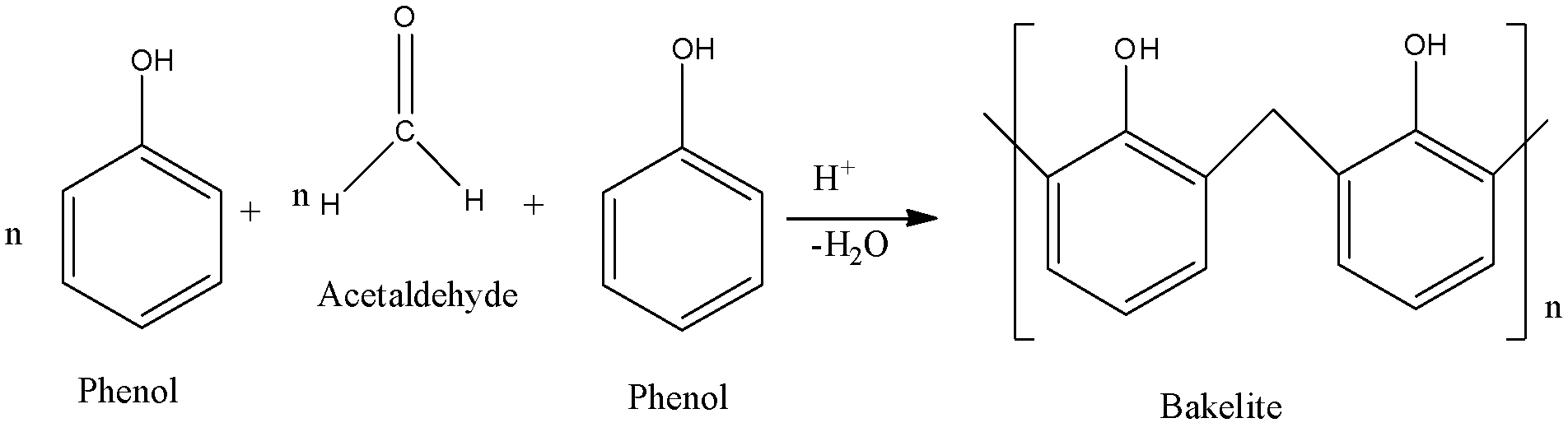
- Bakelite (copolymer of phenol and formaldehyde)
- Buna-N (copolymer of 1,3-butadiene and acrylonitrile )
- Buna-S (copolymer of 1,3-butadiene and styrene )
Therefore, we differentiated between homopolymers and copolymers.
Note: We should also know that there are also some subcategories of copolymers -
- If the two monomers are arranged in an alternative way, it is known as an alternating copolymer.
for example, if the two monomers are P and Q, they will arrange like PQPQPQPQPQPQ
- If the monomers are arranged in a random order like AABAAABBBBAB, it is known as a random copolymer.
- If each monomer joins with the same type of monomers, and then the two blocks of homopolymers join each other. This type is known as block copolymers.
For example, if the two monomers are A and B, they will arrange like AAAAAAABBBBBBB
Complete step by step answer:
We should know that the main difference between copolymer and homopolymer is that copolymers are formed by using two different types of monomers, whereas homopolymers are produced by using a single type of monomer. This difference is responsible for the unique set of properties of homopolymers and copolymers.

- Polythene
- Polytetrafluoroethylene (PTFE or Teflon)
Examples of copolymers:

- Bakelite (copolymer of phenol and formaldehyde)
- Buna-N (copolymer of 1,3-butadiene and acrylonitrile )
- Buna-S (copolymer of 1,3-butadiene and styrene )
Therefore, we differentiated between homopolymers and copolymers.
Note: We should also know that there are also some subcategories of copolymers -
- If the two monomers are arranged in an alternative way, it is known as an alternating copolymer.
for example, if the two monomers are P and Q, they will arrange like PQPQPQPQPQPQ
- If the monomers are arranged in a random order like AABAAABBBBAB, it is known as a random copolymer.
- If each monomer joins with the same type of monomers, and then the two blocks of homopolymers join each other. This type is known as block copolymers.
For example, if the two monomers are A and B, they will arrange like AAAAAAABBBBBBB
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Trending doubts
Which is more stable and why class 12 chemistry CBSE

Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

What are the major means of transport Explain each class 12 social science CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE




