Describe the experiment to compare the E.M.F.’s of two cells by potentiometer under the following headings:
A). Circuit diagram
B). Formula
C). Observation table
D). Two precautions
Answer
624k+ views
Hint: The possible gap between a cell's two terminals can be calculated using a voltmeter, but we can calculate the emf value of a given cell using a potentiometer. Where ${E_1}$ and ${E_2}$ are two-cell EMFs, ${l_1}$ and ${l_2}$ are the lengths for balancing while ${E_1}$ and ${E_2}$, respectively, are attached to the circuit and $\rho $ the potential gradient along the wire of the potentiometer.
Complete step-by-step solution -
Part-A
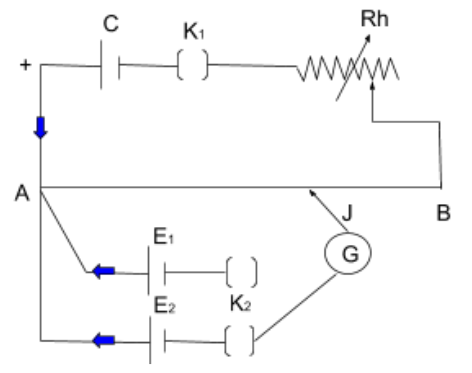
${K_1}$ is the Plug key
${K_2}$ is the Two-way key
$R_h$ is the Rheostat
$J$ is the Jockey
$AB$ is the Potentiometer wire
$G$ is the Galvanometer
${E_1},{E_2}$ are the Experimental/Sample cells
$C$ is the Lead cell
Part-B
Formula:
Let’s consider ${E_1}$ to be the E.M.F. of the first cell.
Let the balancing point that was obtained be at a distance of ${l_1}$. Applying the rule of the principle of potentiometer, we will get-
$ \Rightarrow {E_1} = \rho {l_1}$
Let the above equation be equation 1.
$ \Rightarrow {E_1} = \rho {l_1}$ (equation 1)
Let’s consider ${E_2}$ to be the E.M.F. of the second cell. Let the balancing point be obtained at a distance of ${l_2}$. Thus-
$ \Rightarrow {E_2} = \rho {l_2}$
Let this equation be equation 2.
$ \Rightarrow {E_2} = \rho {l_2}$ (equation 2)
Now, we will divide equation 2 by equation 1, we get-
$ \Rightarrow \dfrac{{{E_1}}}{{{E_2}}} = \dfrac{{\rho {l_1}}}{{\rho {l_2}}} = \dfrac{{{l_1}}}{{{l_2}}}$
Part-C
Observation:
We will observe that the length of the null point for $\left( {\dfrac{{{E_1}}}{{{E_2}}} = \dfrac{{{l_1}}}{{{l_2}}}} \right)$.
${E_1} = {l_1}cm$
${E_2} = {l_2}cm$
Part-D
Precautions:
E.M.F. would be greater than the sample cell of the lead cell.
Each positive terminal should always be connected to only one point.
Note: A potentiometer makes an adjustable voltage divider using a three-terminal resistor with a slipping and rotational contact. It functions as a variable resistor or rheostat if only two terminals are used: one end and the wiper. The measurement device known as potentiometer is simply a voltage divider used to calculate strength (voltage); the variable has the same concept and thus the name of the measuring instrument.
Complete step-by-step solution -
Part-A

${K_1}$ is the Plug key
${K_2}$ is the Two-way key
$R_h$ is the Rheostat
$J$ is the Jockey
$AB$ is the Potentiometer wire
$G$ is the Galvanometer
${E_1},{E_2}$ are the Experimental/Sample cells
$C$ is the Lead cell
Part-B
Formula:
Let’s consider ${E_1}$ to be the E.M.F. of the first cell.
Let the balancing point that was obtained be at a distance of ${l_1}$. Applying the rule of the principle of potentiometer, we will get-
$ \Rightarrow {E_1} = \rho {l_1}$
Let the above equation be equation 1.
$ \Rightarrow {E_1} = \rho {l_1}$ (equation 1)
Let’s consider ${E_2}$ to be the E.M.F. of the second cell. Let the balancing point be obtained at a distance of ${l_2}$. Thus-
$ \Rightarrow {E_2} = \rho {l_2}$
Let this equation be equation 2.
$ \Rightarrow {E_2} = \rho {l_2}$ (equation 2)
Now, we will divide equation 2 by equation 1, we get-
$ \Rightarrow \dfrac{{{E_1}}}{{{E_2}}} = \dfrac{{\rho {l_1}}}{{\rho {l_2}}} = \dfrac{{{l_1}}}{{{l_2}}}$
Part-C
Observation:
We will observe that the length of the null point for $\left( {\dfrac{{{E_1}}}{{{E_2}}} = \dfrac{{{l_1}}}{{{l_2}}}} \right)$.
${E_1} = {l_1}cm$
${E_2} = {l_2}cm$
Part-D
Precautions:
E.M.F. would be greater than the sample cell of the lead cell.
Each positive terminal should always be connected to only one point.
Note: A potentiometer makes an adjustable voltage divider using a three-terminal resistor with a slipping and rotational contact. It functions as a variable resistor or rheostat if only two terminals are used: one end and the wiper. The measurement device known as potentiometer is simply a voltage divider used to calculate strength (voltage); the variable has the same concept and thus the name of the measuring instrument.
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Trending doubts
Which is more stable and why class 12 chemistry CBSE

Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

What are the major means of transport Explain each class 12 social science CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE




