Define the following term:
Zeta potential.
Answer
602.7k+ views
Hint: Zeta potential is also known as Electrokinetic. It is used to define or explain the process of preferential adsorption of ions from solution in the electrical charge on colloidal particles. There are two layers: the fixed layer and the diffused layer.
Complete step by step answer:
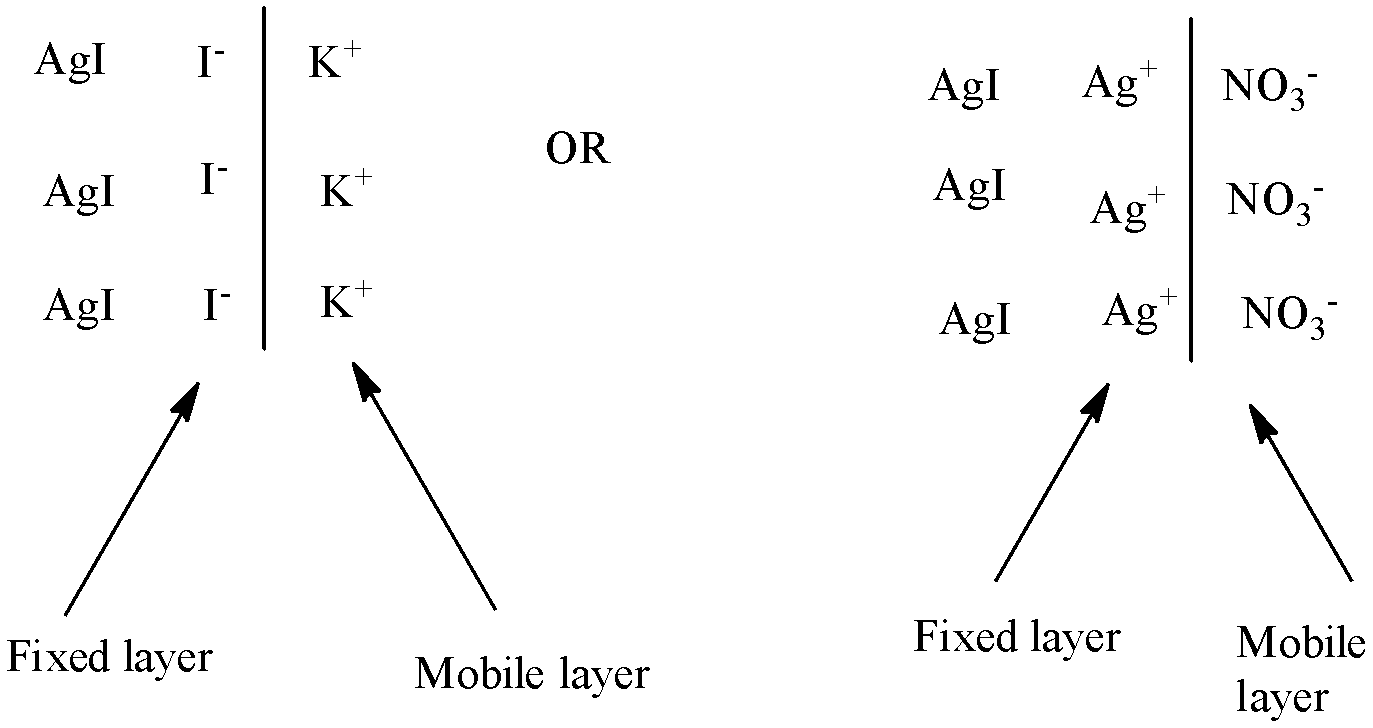
The stability of the colloidal sol is defined by some properties. The electrical charge on the colloidal particle is one of them. The electrical charge on the colloidal particle is explained by the process of preferential adsorption of ions from the solution. During the preparation of colloidal sol, an ionic colloidal adsorb ions common to its lattice.
It can be explained by the colloidal sol $AgI$ is prepared by adding $KI$ solution to the $AgN{{O}_{3}}$ solution till $KI$ is in slight excess, iodide ion (${{I}^{-}}$) will be absorbed in the surface of $AgI$ particles thereby giving them a negative charge:
$AgI+{{I}^{-}}\to AgI:{{I}^{-}}$
The same can be done if the colloidal sol $AgI$ is prepared by adding $AgN{{O}_{3}}$ solution to the $KI$ solution till $AgN{{O}_{3}}$ is in slight excess, iodide ion ($A{{g}^{+}}$ ) will be absorbed in the surface of $AgI$ particles thereby giving them a positive charge:
$AgI+A{{g}^{+}}\to AgI:A{{g}^{+}}$
This can be explained when one type of ions of the electrolyte is adsorbed on the surface of the colloidal particles, there is a formation of a 'fixed layer'. This layer attracts the counter ions from the medium forming a mobile layer known as 'diffused layer'. This potential difference is known as electrokinetic or zeta potential.
Note: Helmholtz electrical double layer is the double layer of opposite charge formed in the zeta potential layer. So, the left-out excess ions remain in the solution thereby giving equal and opposite charge to the dispersion medium.
Complete step by step answer:
The stability of the colloidal sol is defined by some properties. The electrical charge on the colloidal particle is one of them. The electrical charge on the colloidal particle is explained by the process of preferential adsorption of ions from the solution. During the preparation of colloidal sol, an ionic colloidal adsorb ions common to its lattice.
It can be explained by the colloidal sol $AgI$ is prepared by adding $KI$ solution to the $AgN{{O}_{3}}$ solution till $KI$ is in slight excess, iodide ion (${{I}^{-}}$) will be absorbed in the surface of $AgI$ particles thereby giving them a negative charge:
$AgI+{{I}^{-}}\to AgI:{{I}^{-}}$
The same can be done if the colloidal sol $AgI$ is prepared by adding $AgN{{O}_{3}}$ solution to the $KI$ solution till $AgN{{O}_{3}}$ is in slight excess, iodide ion ($A{{g}^{+}}$ ) will be absorbed in the surface of $AgI$ particles thereby giving them a positive charge:
$AgI+A{{g}^{+}}\to AgI:A{{g}^{+}}$
This can be explained when one type of ions of the electrolyte is adsorbed on the surface of the colloidal particles, there is a formation of a 'fixed layer'. This layer attracts the counter ions from the medium forming a mobile layer known as 'diffused layer'. This potential difference is known as electrokinetic or zeta potential.

Note: Helmholtz electrical double layer is the double layer of opposite charge formed in the zeta potential layer. So, the left-out excess ions remain in the solution thereby giving equal and opposite charge to the dispersion medium.
Recently Updated Pages
Three beakers labelled as A B and C each containing 25 mL of water were taken A small amount of NaOH anhydrous CuSO4 and NaCl were added to the beakers A B and C respectively It was observed that there was an increase in the temperature of the solutions contained in beakers A and B whereas in case of beaker C the temperature of the solution falls Which one of the following statements isarecorrect i In beakers A and B exothermic process has occurred ii In beakers A and B endothermic process has occurred iii In beaker C exothermic process has occurred iv In beaker C endothermic process has occurred

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Explain the structure of megasporangium class 12 biology CBSE

What are the major means of transport Explain each class 12 social science CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE




