Conversion of benzene to acetophenone can be brought by:
(a)- Wurtz reaction
(b)- Wurtz fittig reaction
(c)- Friedel craft alkylation
(d)- Friedel craft acylation
Answer
560.6k+ views
Hint: Acetophenone is the compound in which the ketone group is attached to the benzene group. The catalyst used in the reaction is anhydrous aluminum chloride. It is a type of electrophilic substitution reaction.
Complete answer:
Let us study all the options one by one:
(a)- Wurtz reaction:
When an alkyl halide (preferably bromide or iodide ) is treated with metallic sodium in presence of dry ether, symmetrical alkane, containing double the number of carbon atoms present in the alkyl group, is formed.
\[R-X+2Na+X-R\to R-R+2NaX\]
(b)- Wurtz fittig reaction:
Haloarenes are treated with an ethereal solution of an alkyl halide in presence of sodium, form alkyl derivative of benzene.
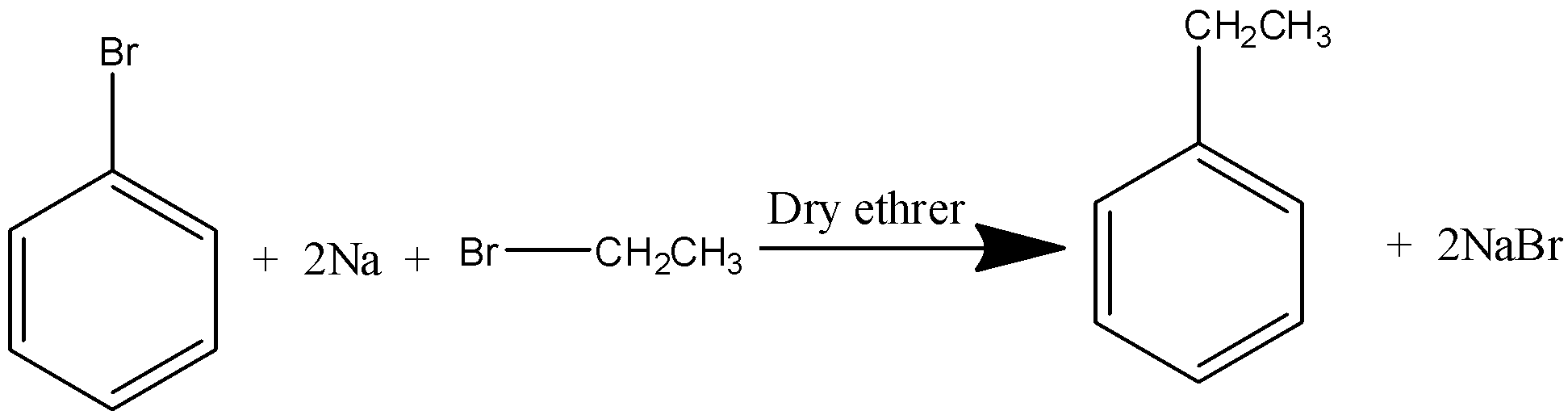
(c)- Friedel craft alkylation:
Benzene reacts with alkyl halides in the presence of a Lewis acid catalyst such as anhydrous aluminum chloride to form alkyl benzene or arenes.
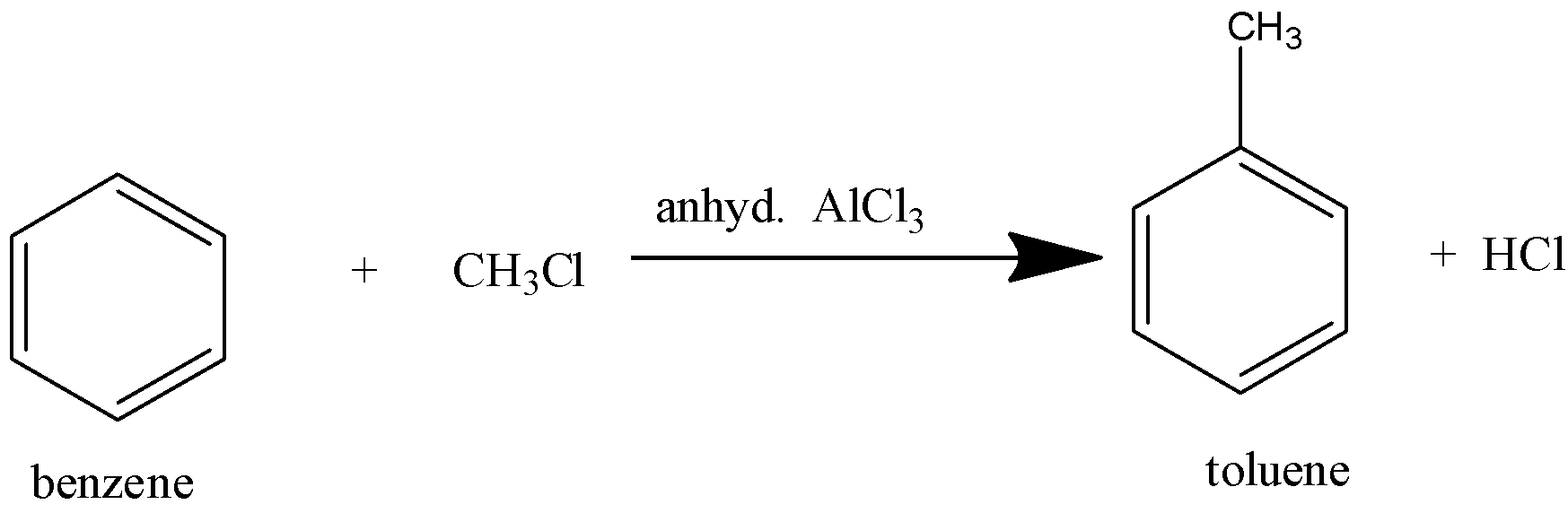
(d)- Friedel craft acylation:
This is one of the most convenient and widely used methods for the preparation of aromatic ketone in which the ketonic group is directly attached to at least one aromatic ring. It involves the treatment of an aromatic hydrocarbon with an acid halide in the presence of a lewis acid-like aluminium chloride.
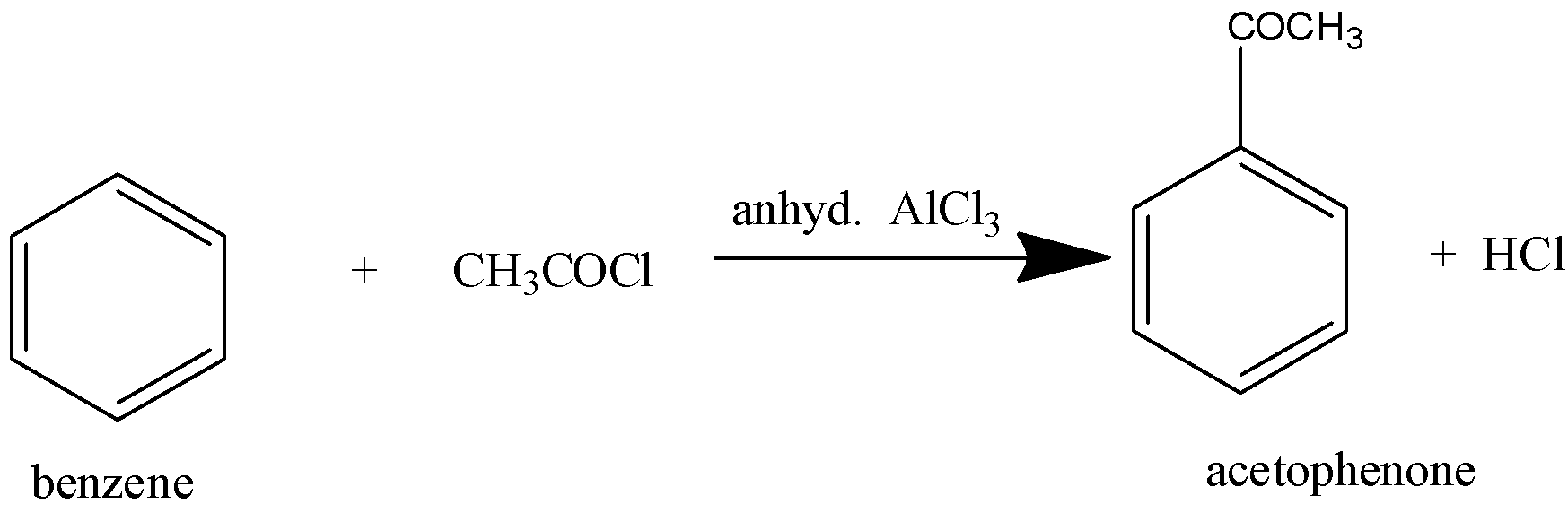
From the above discussion, it can be noted that benzene is converted into acetophenone in Friedel craft acylation reactions.
So, the correct answer is “Option D”.
Note: You may get confused between Friedel craft alkylation and Friedel craft acylation because the catalyst taken is the same but the reactants are only different. Different reactants will yield a different product.
Complete answer:
Let us study all the options one by one:
(a)- Wurtz reaction:
When an alkyl halide (preferably bromide or iodide ) is treated with metallic sodium in presence of dry ether, symmetrical alkane, containing double the number of carbon atoms present in the alkyl group, is formed.
\[R-X+2Na+X-R\to R-R+2NaX\]
(b)- Wurtz fittig reaction:
Haloarenes are treated with an ethereal solution of an alkyl halide in presence of sodium, form alkyl derivative of benzene.

(c)- Friedel craft alkylation:
Benzene reacts with alkyl halides in the presence of a Lewis acid catalyst such as anhydrous aluminum chloride to form alkyl benzene or arenes.

(d)- Friedel craft acylation:
This is one of the most convenient and widely used methods for the preparation of aromatic ketone in which the ketonic group is directly attached to at least one aromatic ring. It involves the treatment of an aromatic hydrocarbon with an acid halide in the presence of a lewis acid-like aluminium chloride.

From the above discussion, it can be noted that benzene is converted into acetophenone in Friedel craft acylation reactions.
So, the correct answer is “Option D”.
Note: You may get confused between Friedel craft alkylation and Friedel craft acylation because the catalyst taken is the same but the reactants are only different. Different reactants will yield a different product.
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Social Science: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

Why is the cell called the structural and functional class 12 biology CBSE

Draw ray diagrams each showing i myopic eye and ii class 12 physics CBSE

Which is the correct genotypic ratio of mendel dihybrid class 12 biology CBSE




