How many constitutional isomers does ${{C}_{3}}{{H}_{8}}O$ have?
Answer
597.6k+ views
Hint: Constitutional isomers are those isomers which have the same number of atoms of each of the elements but differ in the presence of bonds between them that is which is having the distinct bond between them.
Complete step by step answer:
We are familiar with the concepts of organic chemistry which tells us about the types of isomers that can be formed by one molecule and also the variation in their structures or geometries.
Now, we shall see the detailed study about the isomers and its types which also include the constitutional isomers.
- Isomers are the molecules with the identical molecular formula but have the distinct arrangement of atoms in space.
- There are many types of isomers among which constitutional isomer or metamer is one among them which is defined as isomers which have the same number of atoms of each of the elements but are having the distinct bond between them.
- In the given formula, ${{C}_{3}}{{H}_{8}}O$ we can write this in the general formula as ${{C}_{n}}{{H}_{2n-2}}O$, where in if we put n = 3 we get the same formula
- For this type of compound we get two types of functional group that is monohydric alcohol or another possibility is aliphatic ether.
a) The only ether which is possible with this formula will be methoxy ethane that is $C{{H}_{3}}-O-C{{H}_{2}}-C{{H}_{3}}$
b) The monohydric alcohol can be of two forms one is pentan-1-ol which is as shown below,
\[C{{H}_{3}}-C{{H}_{2}}-C{{H}_{2}}-OH\]
The other possibility is the pentan-2-ol which is shown below:
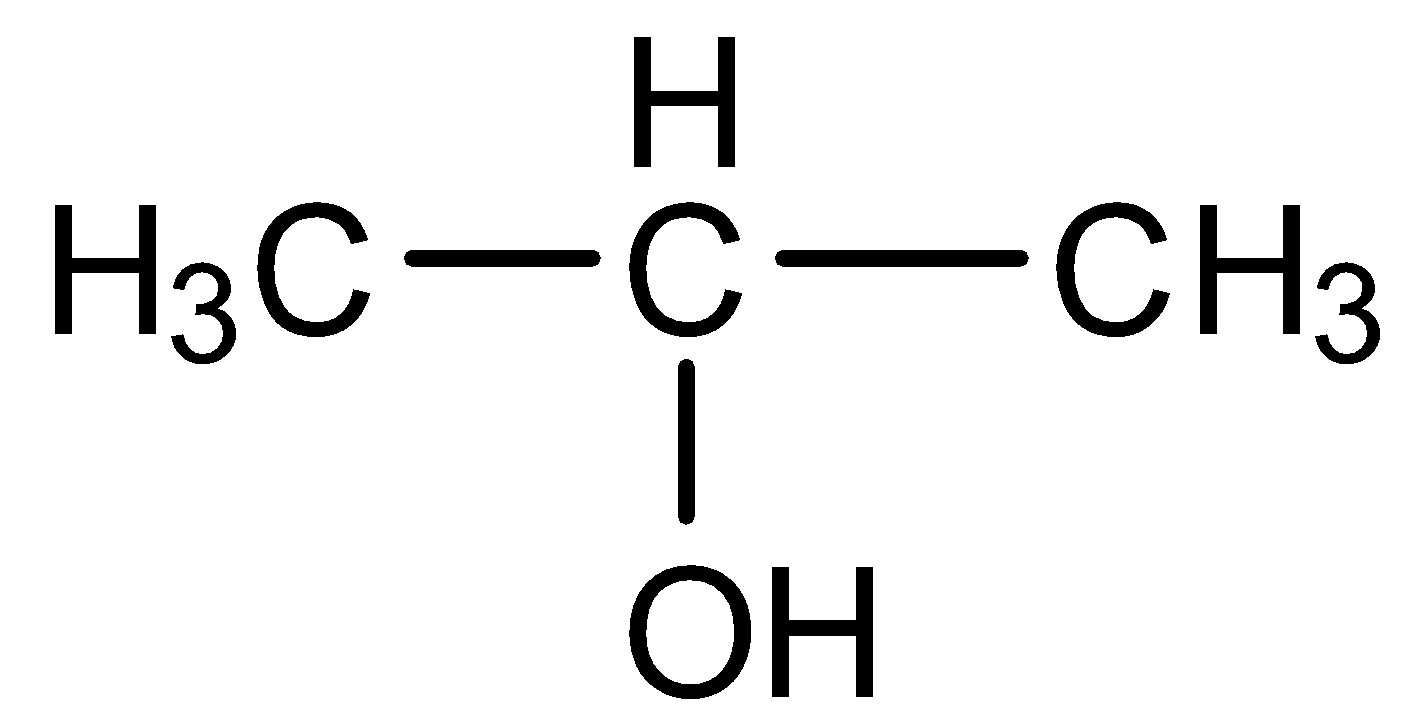
Thus, the correct answer is three constitutional isomers are possible for the molecular formula ${{C}_{3}}{{H}_{8}}O$
Note: Constitutional isomers were also called as metamers formerly which has the same meaning and this concept also applies to the polyatomic ions with the same total charge and the example which can be cited is the cyanate ion.
Complete step by step answer:
We are familiar with the concepts of organic chemistry which tells us about the types of isomers that can be formed by one molecule and also the variation in their structures or geometries.
Now, we shall see the detailed study about the isomers and its types which also include the constitutional isomers.
- Isomers are the molecules with the identical molecular formula but have the distinct arrangement of atoms in space.
- There are many types of isomers among which constitutional isomer or metamer is one among them which is defined as isomers which have the same number of atoms of each of the elements but are having the distinct bond between them.
- In the given formula, ${{C}_{3}}{{H}_{8}}O$ we can write this in the general formula as ${{C}_{n}}{{H}_{2n-2}}O$, where in if we put n = 3 we get the same formula
- For this type of compound we get two types of functional group that is monohydric alcohol or another possibility is aliphatic ether.
a) The only ether which is possible with this formula will be methoxy ethane that is $C{{H}_{3}}-O-C{{H}_{2}}-C{{H}_{3}}$
b) The monohydric alcohol can be of two forms one is pentan-1-ol which is as shown below,
\[C{{H}_{3}}-C{{H}_{2}}-C{{H}_{2}}-OH\]
The other possibility is the pentan-2-ol which is shown below:

Thus, the correct answer is three constitutional isomers are possible for the molecular formula ${{C}_{3}}{{H}_{8}}O$
Note: Constitutional isomers were also called as metamers formerly which has the same meaning and this concept also applies to the polyatomic ions with the same total charge and the example which can be cited is the cyanate ion.
Recently Updated Pages
Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 English: Engaging Questions & Answers for Success

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 9 General Knowledge: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

Why is the cell called the structural and functional class 12 biology CBSE

Draw ray diagrams each showing i myopic eye and ii class 12 physics CBSE

Which is the correct genotypic ratio of mendel dihybrid class 12 biology CBSE




