What is the configuration of the given molecule?
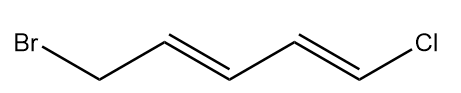
(a) $(1E,\;3Z)$
(b) $(1E,\;3E)$
(c) $(1Z,\;3Z)$
(d) $(1Z,\;3E)$

Answer
527.7k+ views
Hint: The spatial arrangement of atoms in a chiral molecule, and stereochemical description of that atom is known as its absolute configuration. It is determined by CIP rule, according to which the functional group attached to the chiral carbon with higher atomic number will get a greater priority than the functional group with smaller atomic number.
Complete answer:
There are many ways to assign absolute configuration to a molecule. E-Z system is one of the methods according to which, if the groups with same priority are connected to alkene on the same side, then it is said to be Z configuration whereas if the groups with same priority are present on the opposite side of the alkene, then it is termed as E configuration.
In the given molecule, we double bonds at two positions. Let’s look at the configuration of each double bond separately.
Configuration of double bond at first position:
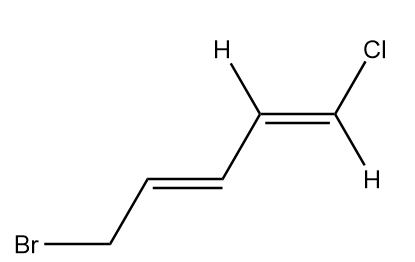
In the molecule, at first terminal chlorine has higher atomic number than hydrogen, so it will be given higher priority whereas in the second terminal of alkene, the carbon atom has higher atomic number than hydrogen, so it will have greater priority.
Therefore, the priorities given are as follows:
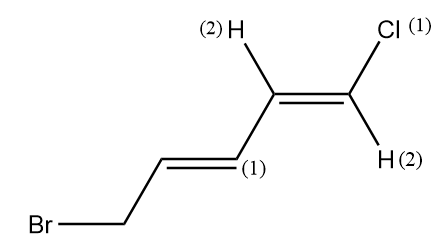
As the groups with the same priority are present at the opposite sides of the alkene, therefore it has E configuration.
Configuration of double bond at third position:
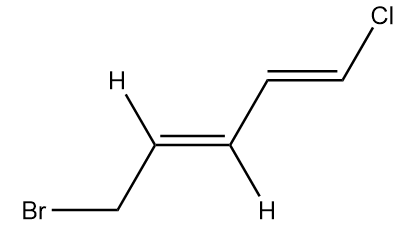
In the molecule, at first the carbon atom has higher atomic number than hydrogen, so it will be given higher priority whereas in the second terminal of alkene, again the carbon atom has higher atomic number than hydrogen, so it will have greater priority.
Therefore, the priorities given are as follows:
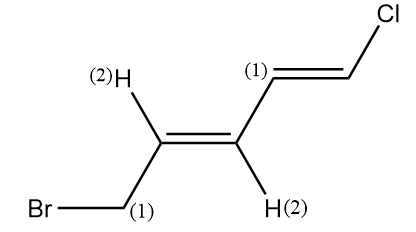
As the groups with the same priority are present at the opposite sides of the alkene, therefore it has E configuration.
Hence, for the given molecule the absolute configuration is $(1E,\,3E)$.
Therefore, the correct answer is option B.
Note:
Other than E-Z configuration, the other absolute configurations which are based on CIP rules are R-S configuration that is an important nomenclature for denoting enantiomers and d-l configuration that is determined by a direction in which an optically active compound rotates a plane of polarized light.
Complete answer:
There are many ways to assign absolute configuration to a molecule. E-Z system is one of the methods according to which, if the groups with same priority are connected to alkene on the same side, then it is said to be Z configuration whereas if the groups with same priority are present on the opposite side of the alkene, then it is termed as E configuration.
In the given molecule, we double bonds at two positions. Let’s look at the configuration of each double bond separately.
Configuration of double bond at first position:

In the molecule, at first terminal chlorine has higher atomic number than hydrogen, so it will be given higher priority whereas in the second terminal of alkene, the carbon atom has higher atomic number than hydrogen, so it will have greater priority.
Therefore, the priorities given are as follows:

As the groups with the same priority are present at the opposite sides of the alkene, therefore it has E configuration.
Configuration of double bond at third position:

In the molecule, at first the carbon atom has higher atomic number than hydrogen, so it will be given higher priority whereas in the second terminal of alkene, again the carbon atom has higher atomic number than hydrogen, so it will have greater priority.
Therefore, the priorities given are as follows:

As the groups with the same priority are present at the opposite sides of the alkene, therefore it has E configuration.
Hence, for the given molecule the absolute configuration is $(1E,\,3E)$.
Therefore, the correct answer is option B.
Note:
Other than E-Z configuration, the other absolute configurations which are based on CIP rules are R-S configuration that is an important nomenclature for denoting enantiomers and d-l configuration that is determined by a direction in which an optically active compound rotates a plane of polarized light.
Recently Updated Pages
Three beakers labelled as A B and C each containing 25 mL of water were taken A small amount of NaOH anhydrous CuSO4 and NaCl were added to the beakers A B and C respectively It was observed that there was an increase in the temperature of the solutions contained in beakers A and B whereas in case of beaker C the temperature of the solution falls Which one of the following statements isarecorrect i In beakers A and B exothermic process has occurred ii In beakers A and B endothermic process has occurred iii In beaker C exothermic process has occurred iv In beaker C endothermic process has occurred

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

What are the major means of transport Explain each class 12 social science CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE

Why should a magnesium ribbon be cleaned before burning class 12 chemistry CBSE




