Choose the correct answer among the alternatives given:
Classify the following compounds as primary, secondary and tertiary halides.
(i) 1-Bromobut-2-ene
(ii) 4-Bromopent-2-ene
(iii) 2-Bromo-2-methylpropane
(A) (i)-secondary, (ii)-tertiary and (iii)-primary
(B) (i)-secondary, (ii)-primary and (iii)-tertiary
(C) (i)-primary, (ii)-tertiary and (iii)-secondary
(D) (i)-primary, (ii)-secondary and (iii)-tertiary
Answer
613.5k+ views
Hint: In primary halides, the halogen atom is bonded to a primary carbon atom. In secondary halides, the halogen atom is bonded to a secondary carbon atom while in tertiary halide; the halogen atom is bonded to a tertiary carbon atom.
Complete step by step answer:
In order to solve this question, we need to first understand the meaning of primary, secondary and tertiary halides.
Primary Halides: In primary halides, the carbon atom bonded to the halogen atom is bonded to only one carbon atom and the rest are hydrogen atoms or the carbon bonded to the halogen atom is only bonded to hydrogen atoms. More simply, we can say that the halogen atom is bonded to a primary carbon.
Example:
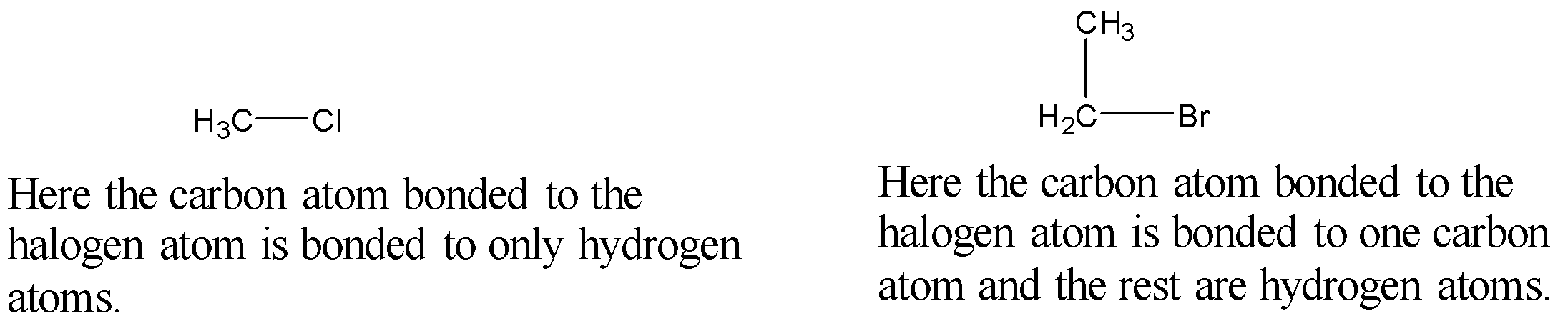
Secondary Halides: In secondary halides, the carbon atom bonded to the halogen atom is bonded to two carbon atoms and one hydrogen atom. More simply, we can say that the halogen atom is bonded to a secondary carbon.
Example:
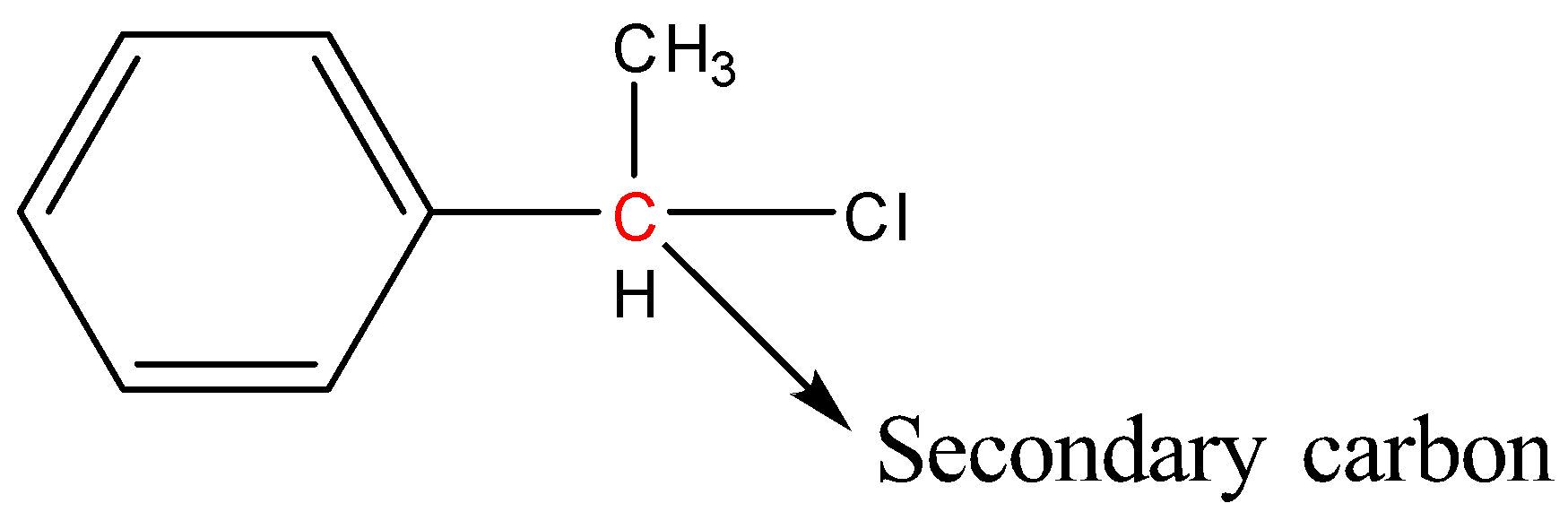
Tertiary halides: In tertiary halides, the carbon atom bonded to the halogen atom is bonded to three carbon atoms. More simply we can say that the halogen atom is bonded to a tertiary carbon.
Example:
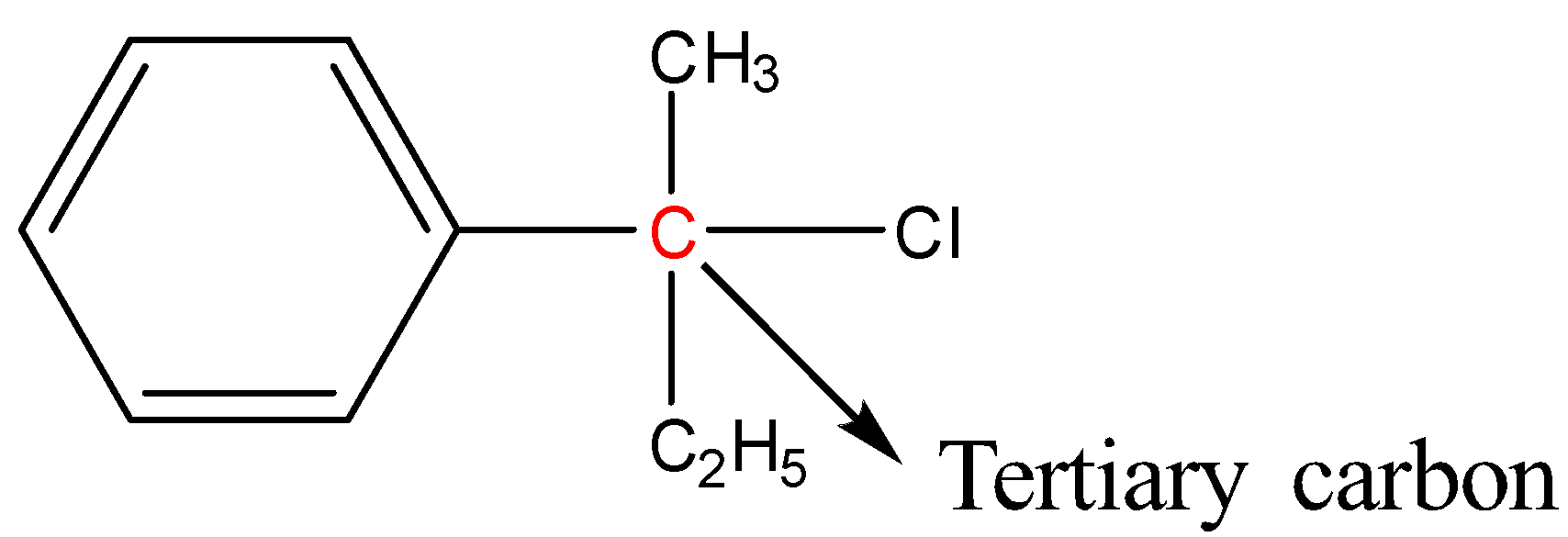
In order to solve this question, we need to draw the structures of the compounds given:
1-Bromobut-2-ene
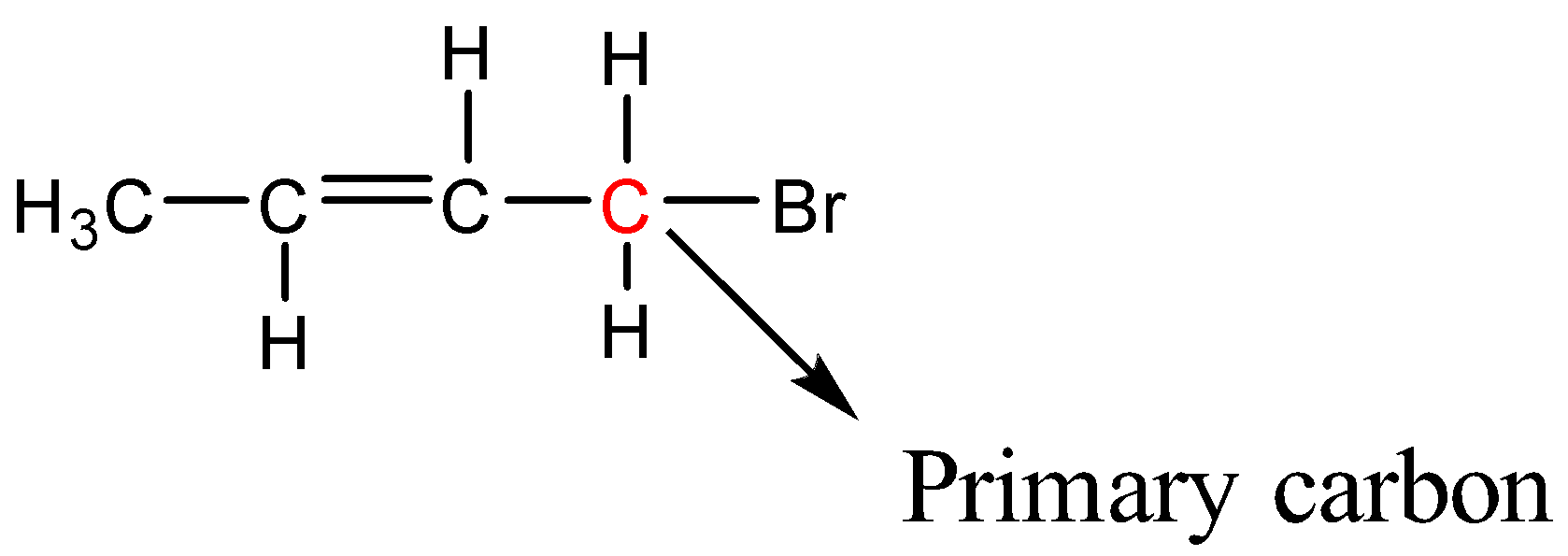
Here Bromine is bonded to a primary carbon atom; therefore 1-Bromobut-2-ene is a primary halide.
4-Bromopent-2-ene
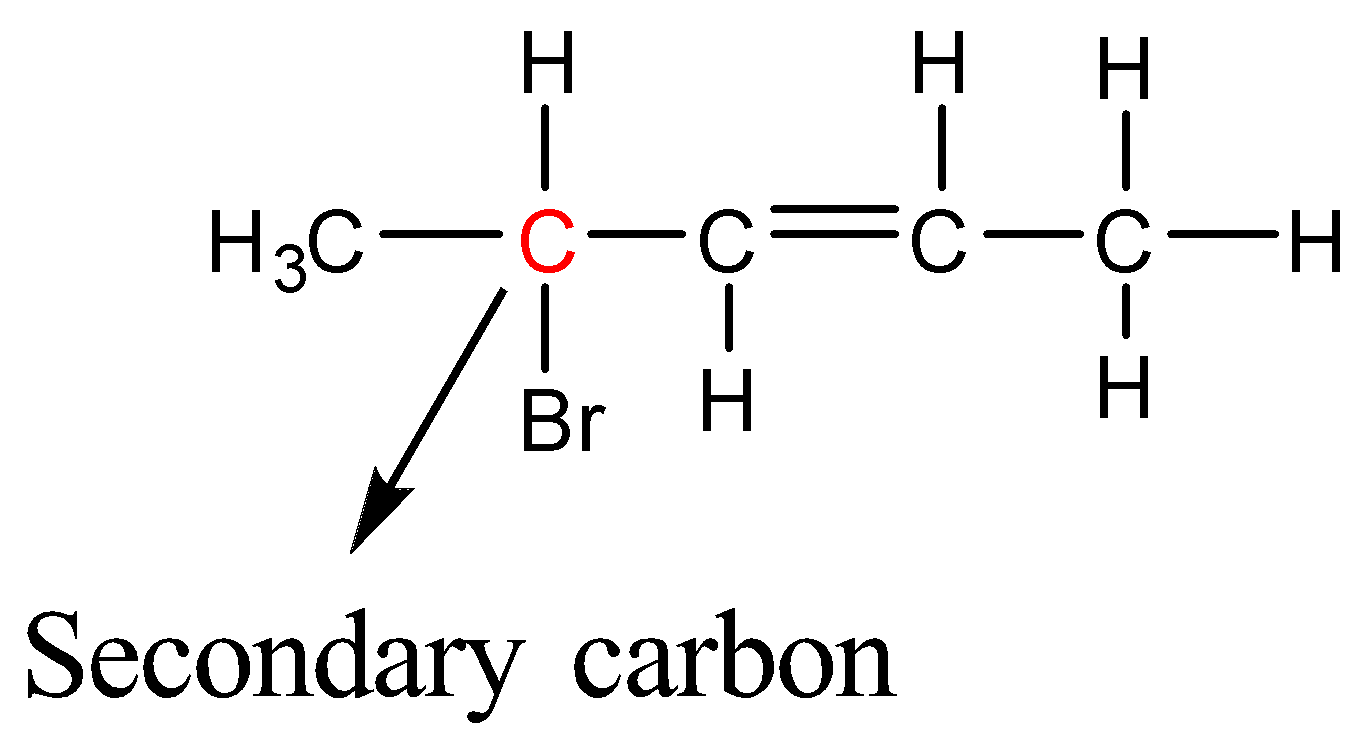
Here Bromine is bonded to a secondary carbon atom; therefore 4-Bromopent-2-ene is a secondary halide.
2-Bromo-2-methylpropane
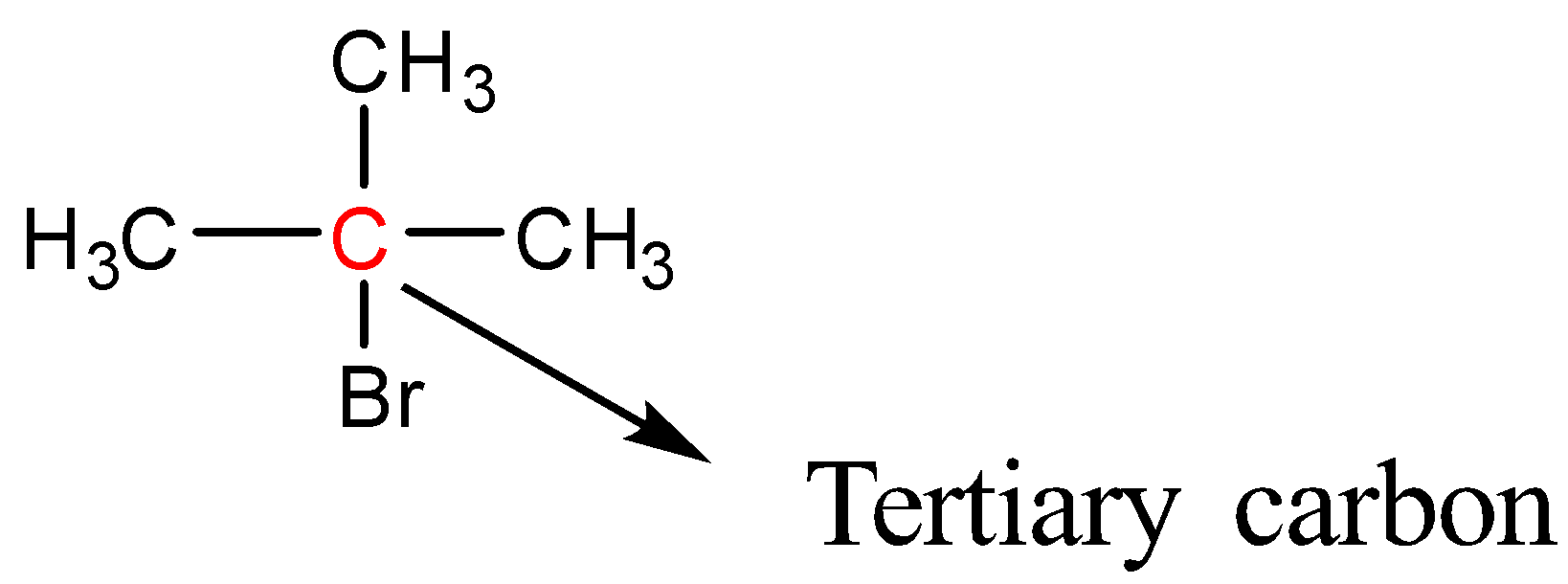
Here Bromine is bonded to a tertiary carbon atom; therefore 2-Bromo-2-methylpropane is a tertiary halide. So, the correct answer is “Option D”.
Note: In this question all the names of the organic compound were given in accordance with the IUPAC (International Union of Pure and Applied Chemistry) nomenclature. Therefore in order to draw the structure of the compounds from their names, you should know the rules of the IUPAC nomenclature.
Complete step by step answer:
In order to solve this question, we need to first understand the meaning of primary, secondary and tertiary halides.
Primary Halides: In primary halides, the carbon atom bonded to the halogen atom is bonded to only one carbon atom and the rest are hydrogen atoms or the carbon bonded to the halogen atom is only bonded to hydrogen atoms. More simply, we can say that the halogen atom is bonded to a primary carbon.
Example:
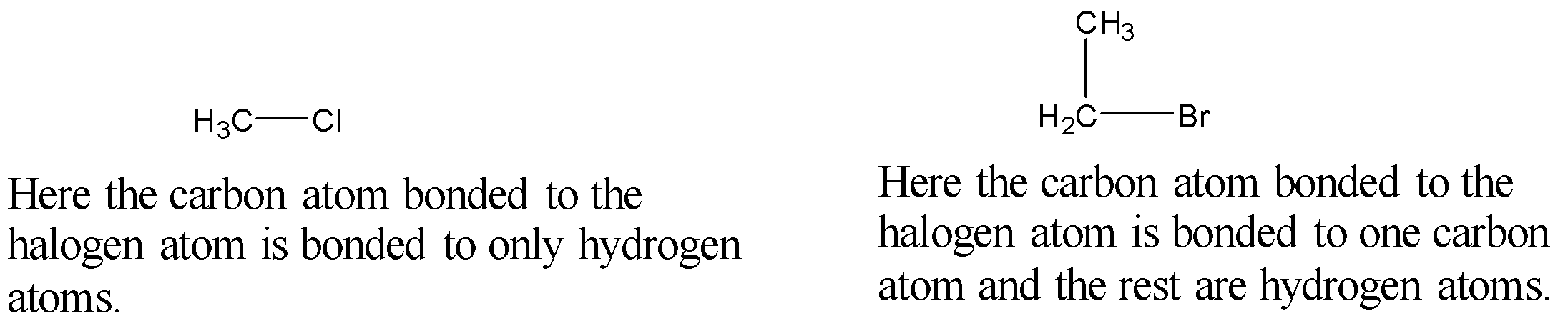
Secondary Halides: In secondary halides, the carbon atom bonded to the halogen atom is bonded to two carbon atoms and one hydrogen atom. More simply, we can say that the halogen atom is bonded to a secondary carbon.
Example:

Tertiary halides: In tertiary halides, the carbon atom bonded to the halogen atom is bonded to three carbon atoms. More simply we can say that the halogen atom is bonded to a tertiary carbon.
Example:

In order to solve this question, we need to draw the structures of the compounds given:
1-Bromobut-2-ene

Here Bromine is bonded to a primary carbon atom; therefore 1-Bromobut-2-ene is a primary halide.
4-Bromopent-2-ene

Here Bromine is bonded to a secondary carbon atom; therefore 4-Bromopent-2-ene is a secondary halide.
2-Bromo-2-methylpropane

Here Bromine is bonded to a tertiary carbon atom; therefore 2-Bromo-2-methylpropane is a tertiary halide. So, the correct answer is “Option D”.
Note: In this question all the names of the organic compound were given in accordance with the IUPAC (International Union of Pure and Applied Chemistry) nomenclature. Therefore in order to draw the structure of the compounds from their names, you should know the rules of the IUPAC nomenclature.
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Trending doubts
Which is more stable and why class 12 chemistry CBSE

Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

What are the major means of transport Explain each class 12 social science CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE




