How many chiral carbon atoms are there in \[1-\] chloro \[-2,5-\] dimethyl cyclohexane?
A. $3$
B. $1$
C. $2$
D. $0$
Answer
595.8k+ views
Hint: Chiral compounds are the compounds that cannot be superimposed on its mirror image by the combination of rotation. Chiral compounds exist in two isomers that show mirror images of each other. These are also enantiomers.
Complete answer:
Before talking about the answer, we should know what a chiral molecule is. A chiral molecule consists of a chiral center. When this center coincides with another atom, that substance is said to have point chirality. A compound is said to be chiral when there is no plane of symmetry and center of symmetry. If it does not have either of these, then it is said to be chiral.
Plane of symmetry is defined as a plane that divides a molecule in two halves that shows mirror images of each other.
In this question, \[1-\] chloro \[-2,5-\] dimethyl cyclohexane has two planes of separation, that means, the either side of the plane is different with carbon having different groups. One chiral carbon atom is attached with chlorine, the second chiral carbon is attached to methyl group and the third chiral carbon atom is attached with methyl group that does not contain any neighbor groups.
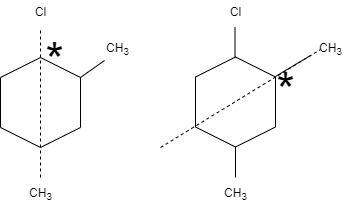
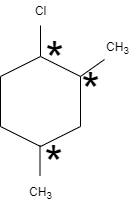
Therefore, \[1-\] chloro \[-2,5-\] dimethyl cyclohexane contains three chiral carbon atoms.
Hence, the correct option is (A) $3$
Additional information:
-As we have discussed that chiral compounds are the compounds that do not show superimposable mirror images, whereas achiral compounds are the compounds that show superimposable mirror images of each other.
-Chiral compounds do not show planes of symmetry or center of symmetry, whereas achiral compounds show planes of symmetry or center of symmetry.
Note: Center of symmetry is defined as a point from where if we move towards a group, it shows equal distance when we move in the opposite direction of the equivalent group.
A molecule shows a center of symmetry only when identical atoms exist in that molecule.
Complete answer:
Before talking about the answer, we should know what a chiral molecule is. A chiral molecule consists of a chiral center. When this center coincides with another atom, that substance is said to have point chirality. A compound is said to be chiral when there is no plane of symmetry and center of symmetry. If it does not have either of these, then it is said to be chiral.
Plane of symmetry is defined as a plane that divides a molecule in two halves that shows mirror images of each other.
In this question, \[1-\] chloro \[-2,5-\] dimethyl cyclohexane has two planes of separation, that means, the either side of the plane is different with carbon having different groups. One chiral carbon atom is attached with chlorine, the second chiral carbon is attached to methyl group and the third chiral carbon atom is attached with methyl group that does not contain any neighbor groups.


Therefore, \[1-\] chloro \[-2,5-\] dimethyl cyclohexane contains three chiral carbon atoms.
Hence, the correct option is (A) $3$
Additional information:
-As we have discussed that chiral compounds are the compounds that do not show superimposable mirror images, whereas achiral compounds are the compounds that show superimposable mirror images of each other.
-Chiral compounds do not show planes of symmetry or center of symmetry, whereas achiral compounds show planes of symmetry or center of symmetry.
Note: Center of symmetry is defined as a point from where if we move towards a group, it shows equal distance when we move in the opposite direction of the equivalent group.
A molecule shows a center of symmetry only when identical atoms exist in that molecule.
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Trending doubts
Which is more stable and why class 12 chemistry CBSE

Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

What are the major means of transport Explain each class 12 social science CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE




