What is the charge on the polyatomic ions nitrite and chlorite?
Answer
545.7k+ views
Hint :We know that for solving this problem, you should have the knowledge about polyatomic ion and oxidation number.
A Polyatomic ion is an ion containing more than one atom. Oxidation number is the number of electrons lost or gained by an element during a reaction. Charges with respect to the Lewis structure, the charge on a polyatomic ion can be identified easily.
Complete Step By Step Answer:
Ions are those molecules or compounds that have either a positive or negative charge. They are classified as monatomic in which only one atom is present with either positive or negative charge, diatomic in which two atoms are present, and polyatomic in which more than two atoms are present. Lewis structure helps in representing valence electrons of a molecule. On calculating the formal.
Step-1: To calculate the charge on a polyatomic ion, the oxidation number of all the atoms can be added together.
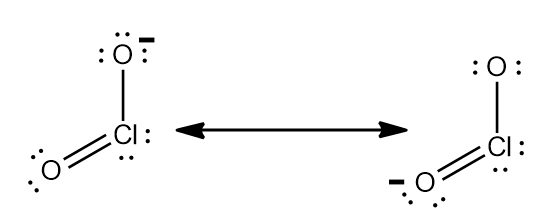
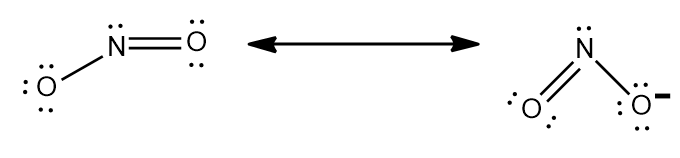
Step-2: Nitrite ion stands for $ NO_{2}^{-} $ .So, charge can be calculated as sum of oxidation number of nitrogen and oxygen.
Similarly Chlorite ion stands for $ ClO_{2}^{-} $ .
Step-3: The charge can also be calculated using Lewis structure and making sure every atom in the ion has $ 8 $
electrons. Using the given phenomenon and studying the Lewis structure of $ NO_{2}^{-} $ and $ ClO_{2}^{-} $ ,they both
$ -1 $ charge.
Note :
There are many ways to identify the charge on a polyatomic ion and one can use any way they find easy to remember. But making the correct Lewis structure is an important step which must not be forgotten. The overall ionic formula of the compound must be electrically neutral, that is it should have zero charges and while writing the formula of the compound we assure that the cation part should come first followed by anion.
A Polyatomic ion is an ion containing more than one atom. Oxidation number is the number of electrons lost or gained by an element during a reaction. Charges with respect to the Lewis structure, the charge on a polyatomic ion can be identified easily.
Complete Step By Step Answer:
Ions are those molecules or compounds that have either a positive or negative charge. They are classified as monatomic in which only one atom is present with either positive or negative charge, diatomic in which two atoms are present, and polyatomic in which more than two atoms are present. Lewis structure helps in representing valence electrons of a molecule. On calculating the formal.
Step-1: To calculate the charge on a polyatomic ion, the oxidation number of all the atoms can be added together.


Step-2: Nitrite ion stands for $ NO_{2}^{-} $ .So, charge can be calculated as sum of oxidation number of nitrogen and oxygen.
Similarly Chlorite ion stands for $ ClO_{2}^{-} $ .
Step-3: The charge can also be calculated using Lewis structure and making sure every atom in the ion has $ 8 $
electrons. Using the given phenomenon and studying the Lewis structure of $ NO_{2}^{-} $ and $ ClO_{2}^{-} $ ,they both
$ -1 $ charge.
Note :
There are many ways to identify the charge on a polyatomic ion and one can use any way they find easy to remember. But making the correct Lewis structure is an important step which must not be forgotten. The overall ionic formula of the compound must be electrically neutral, that is it should have zero charges and while writing the formula of the compound we assure that the cation part should come first followed by anion.
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Trending doubts
Which is more stable and why class 12 chemistry CBSE

Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

What are the major means of transport Explain each class 12 social science CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE




