Calculate the potential $E$ for $F{{e}^{3+}}|F{{e}^{2+}}$ electrode when the concentration of $F{{e}^{2+}}$ is exactly five times that of $F{{e}^{3+}}$.
Answer
530.7k+ views
Hint: To find the potential for the given electrode i.e., $F{{e}^{3+}}|F{{e}^{2+}}$, first determine the standard electrode potential from the datasheet and with the help of Nernst equation, we can calculate the potential for the given electrode.
Formula used-
$E={{E}^{o}}-\dfrac{0.059}{n}\log \dfrac{[P]}{[R]}$
Where, ${{E}^{o}}$ is the standard electrode potential of the electrode, n is the change in number of electrons during the reaction, $[P]$ is the concentration of ion in product and $[R]$ is the concentration of ion in reactant.
Complete answer:
The value of standard EMF of a cell or potential of the cell when the concentration of species is considered as unity is known as standard electrode potential and it is denoted by ${{E}^{o}}$. Some basic elements along with their standard electrode potential are given as per following table-
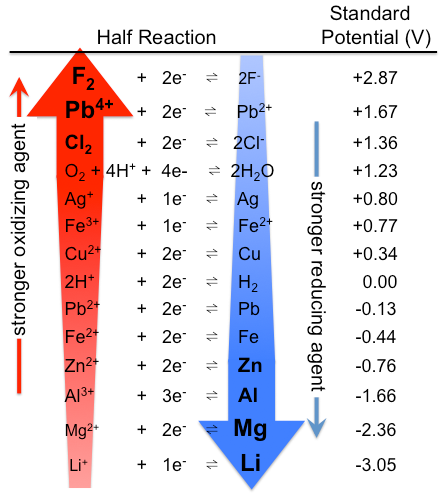
Now, the $F{{e}^{3+}}|F{{e}^{2+}}$ electrode can be represented by the following equation:
$F{{e}^{3+}}+{{e}^{-}}\to F{{e}^{2+}}$
And as per given table, the value for standard electrode potential for the given electrode $E_{F{{e}^{3+}}|F{{e}^{2+}}}^{o}=0.77V$. We know that the Nernst equation is used to relate reduction potentials to standard electrode potential, temperature and concentration of the species undergoing reduction and oxidation. So, according to the Nernst equation:
$E=E_{F{{e}^{3+}}|F{{e}^{2+}}}^{o}-\dfrac{0.059}{n}\log \dfrac{\left[ F{{e}^{2+}} \right]}{\left[ F{{e}^{3+}} \right]}$
It is given in the question that the concentration of $F{{e}^{2+}}$ is exactly five times that of $F{{e}^{3+}}$ which means $\left[ F{{e}^{2+}} \right]=5\times \left[ F{{e}^{3+}} \right]$. Substituting values in the equation:
$\Rightarrow E=0.77-\dfrac{0.059}{1}\log \dfrac{5\times \left[ F{{e}^{3+}} \right]}{\left[ F{{e}^{3+}} \right]}$
$\Rightarrow E=0.77-0.059\log 5$
$\Rightarrow E=0.73V$
Hence, the potential for $F{{e}^{3+}}|F{{e}^{2+}}$ electrode under given conditions is $0.73V$.
Note:
It is important to note that in this question, the formula used for the Nernst equation is only applicable when the temperature of the reaction is taken as ${{25}^{o}}C$. As no temperature was mentioned in the question, so here we calculated the potential at room temperature i.e., ${{25}^{o}}C$. In the case if temperature (other than room temperature) is given then the Nernst equation to be used is $E={{E}^{o}}-\dfrac{RT}{nF}\log \dfrac{[P]}{[R]}$.
Formula used-
$E={{E}^{o}}-\dfrac{0.059}{n}\log \dfrac{[P]}{[R]}$
Where, ${{E}^{o}}$ is the standard electrode potential of the electrode, n is the change in number of electrons during the reaction, $[P]$ is the concentration of ion in product and $[R]$ is the concentration of ion in reactant.
Complete answer:
The value of standard EMF of a cell or potential of the cell when the concentration of species is considered as unity is known as standard electrode potential and it is denoted by ${{E}^{o}}$. Some basic elements along with their standard electrode potential are given as per following table-

Now, the $F{{e}^{3+}}|F{{e}^{2+}}$ electrode can be represented by the following equation:
$F{{e}^{3+}}+{{e}^{-}}\to F{{e}^{2+}}$
And as per given table, the value for standard electrode potential for the given electrode $E_{F{{e}^{3+}}|F{{e}^{2+}}}^{o}=0.77V$. We know that the Nernst equation is used to relate reduction potentials to standard electrode potential, temperature and concentration of the species undergoing reduction and oxidation. So, according to the Nernst equation:
$E=E_{F{{e}^{3+}}|F{{e}^{2+}}}^{o}-\dfrac{0.059}{n}\log \dfrac{\left[ F{{e}^{2+}} \right]}{\left[ F{{e}^{3+}} \right]}$
It is given in the question that the concentration of $F{{e}^{2+}}$ is exactly five times that of $F{{e}^{3+}}$ which means $\left[ F{{e}^{2+}} \right]=5\times \left[ F{{e}^{3+}} \right]$. Substituting values in the equation:
$\Rightarrow E=0.77-\dfrac{0.059}{1}\log \dfrac{5\times \left[ F{{e}^{3+}} \right]}{\left[ F{{e}^{3+}} \right]}$
$\Rightarrow E=0.77-0.059\log 5$
$\Rightarrow E=0.73V$
Hence, the potential for $F{{e}^{3+}}|F{{e}^{2+}}$ electrode under given conditions is $0.73V$.
Note:
It is important to note that in this question, the formula used for the Nernst equation is only applicable when the temperature of the reaction is taken as ${{25}^{o}}C$. As no temperature was mentioned in the question, so here we calculated the potential at room temperature i.e., ${{25}^{o}}C$. In the case if temperature (other than room temperature) is given then the Nernst equation to be used is $E={{E}^{o}}-\dfrac{RT}{nF}\log \dfrac{[P]}{[R]}$.
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Social Science: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

In order to find out the different types of gametes class 12 biology NEET_UG

Why is the cell called the structural and functional class 12 biology CBSE

Draw ray diagrams each showing i myopic eye and ii class 12 physics CBSE




