${{C}_{6}}{{H}_{5}}S{{O}_{3}}H\xrightarrow{{{H}_{2}}O}A\xrightarrow{{{H}_{2}}/Ni}B$, then A and B respectively:
(A) ${{C}_{6}}{{H}_{6}}$, Cyclohexane
(B) ${{C}_{6}}{{H}_{6}}$, Cyclohexene
(C) ${{C}_{6}}{{H}_{6}}$, Cyclohexadiene
(D) ${{C}_{6}}{{H}_{5}}OH$, Cyclohexane
Answer
584.7k+ views
Hint: The reactant undergoing the change is benzene sulphonic acid.
- The mixture of hydrogen along with Ni is the reagent used for hydrogenation.
Complete step by step answer:
So in the question a chemical reaction is given and we have to predict the products formed in each step of the reaction. From the time we are studying organic compounds we have gone through many reagents and many conversion mechanisms, let's brush up a few reactions from our memory.
- So first identify the compound which is undergoing the change. The reactant here is the benzene sulphonic acid and it is getting attacked by water.
Benzene sulphonic acid is a very strong acid which readily gets attacked by water and undergoes the desulfonylation group ie the sulphonyl group-$-S{{O}_{3}}H$ is getting removed from the molecule.
- So to undergo desulfonylation the required condition is that the reaction must be done in the presence of high concentration of water and heat must be supplied for the reaction with very minimal concentration of sulphuric or say any general acids.
So let’s trace the mechanism of the reaction:
- In the desulfonylation reaction first the water which is a weak base attacks the –OH group of the sulfonyl groups and abstracts the proton and forms the conjugate base of benzene sulphonic acid. And then the double bond in the benzene acts as the nucleophile and attacks the ${{H}_{3}}{{O}^{+}}$ and in the next step the heat is the very required element since at this step the sulphonyl group is removed as $S{{O}_{3}}$.
The mechanism of the reaction is as follows:
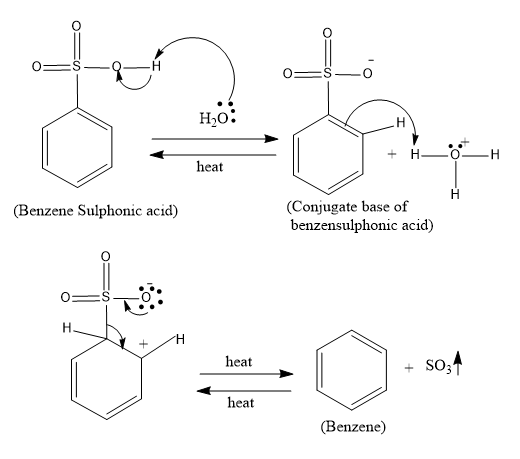
So the overall reaction is as follows:
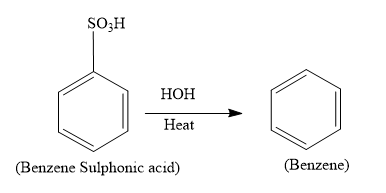
Hence the product (A) formed is benzene which has the formulae, ${{C}_{6}}{{H}_{6}}$
- Now let’s move to the next section, here the reagent used is hydrogen and Ni.As we are very familiar that this mixture is used for the hydrogenation of a molecule ie to add on hydrogen to the multiple bonds or we can say that the multiple bonds are getting reduced.
So just add the hydrogen atom in between the double bonds.
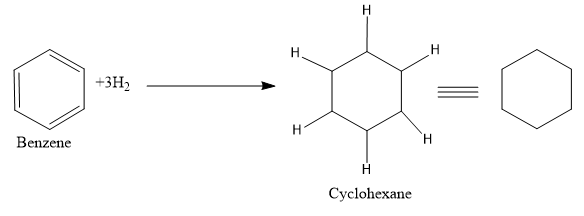
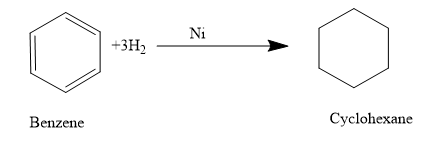
So the product (B) formed is cyclohexane The correct answer is option “A” .
Additional Information :
Note: The reaction is the reverse reaction of the sulfonation reaction of benzene molecules. In the sulphonation of benzene molecules we treat the molecule with sulphuric acid to yield benzene sulphonic acid as the final product.
The other hydrogenation reagent used is $Pd/C$ with ${{H}_{2}}$
- The mixture of hydrogen along with Ni is the reagent used for hydrogenation.
Complete step by step answer:
So in the question a chemical reaction is given and we have to predict the products formed in each step of the reaction. From the time we are studying organic compounds we have gone through many reagents and many conversion mechanisms, let's brush up a few reactions from our memory.
- So first identify the compound which is undergoing the change. The reactant here is the benzene sulphonic acid and it is getting attacked by water.
Benzene sulphonic acid is a very strong acid which readily gets attacked by water and undergoes the desulfonylation group ie the sulphonyl group-$-S{{O}_{3}}H$ is getting removed from the molecule.
- So to undergo desulfonylation the required condition is that the reaction must be done in the presence of high concentration of water and heat must be supplied for the reaction with very minimal concentration of sulphuric or say any general acids.
So let’s trace the mechanism of the reaction:
- In the desulfonylation reaction first the water which is a weak base attacks the –OH group of the sulfonyl groups and abstracts the proton and forms the conjugate base of benzene sulphonic acid. And then the double bond in the benzene acts as the nucleophile and attacks the ${{H}_{3}}{{O}^{+}}$ and in the next step the heat is the very required element since at this step the sulphonyl group is removed as $S{{O}_{3}}$.
The mechanism of the reaction is as follows:

So the overall reaction is as follows:

Hence the product (A) formed is benzene which has the formulae, ${{C}_{6}}{{H}_{6}}$
- Now let’s move to the next section, here the reagent used is hydrogen and Ni.As we are very familiar that this mixture is used for the hydrogenation of a molecule ie to add on hydrogen to the multiple bonds or we can say that the multiple bonds are getting reduced.
So just add the hydrogen atom in between the double bonds.


So the product (B) formed is cyclohexane The correct answer is option “A” .
Additional Information :
Note: The reaction is the reverse reaction of the sulfonation reaction of benzene molecules. In the sulphonation of benzene molecules we treat the molecule with sulphuric acid to yield benzene sulphonic acid as the final product.
The other hydrogenation reagent used is $Pd/C$ with ${{H}_{2}}$
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Trending doubts
Which is more stable and why class 12 chemistry CBSE

Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

What are the major means of transport Explain each class 12 social science CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE




