How will you bring conversion of isopropyl chloride to n-propyl chloride$?$
Answer
554.1k+ views
Hint: Haloalkanes containing $\beta $- hydrogen atoms undergo elimination reaction in presence of alcoholic KOH. Such reactions are known as dehydrohalogenation reactions or $\beta $elimination reactions.
Complete answer:
-When haloalkanes having $\beta $-hydrogen atoms are heated in the presence of alcoholic solution of potassium hydroxide, then they undergo elimination reaction. Such reactions are also known as $\beta $- elimination reactions and results in the formation of alkenes.
-These reactions are known as $\beta $- elimination reactions because the hydrogen present at the $\beta $position of the alkanes is removed. The hydrogen atom which is attached to the carbon atom present next to that carbon atom which carries halogen is called $\beta $- hydrogen.
-To convert isopropyl chloride to n-propyl chloride first treat isopropyl chloride with alcoholic KOH to form prop$-1-$ene and eliminate HCl.
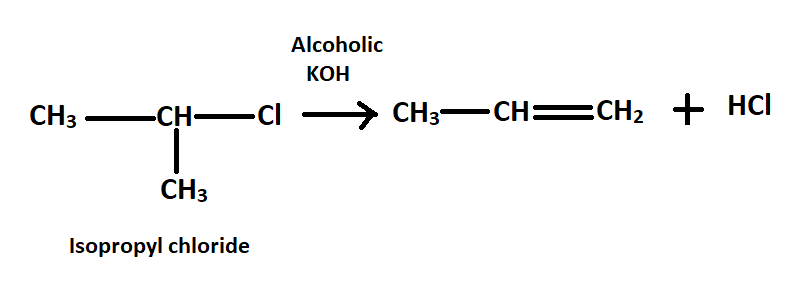
-When boiled with alcoholic KOH, the hydrogen atom transfers its electron pair to the adjacent carbon atom and the bromide gets removed. This forms a double bond between the $\alpha $ and $\beta $carbon atom.
- During the reaction halogen gets eliminated with hydrogen as hydrogen halide and thus alkene is formed.
-Then this HCl is treated with prop$-1-$ene in presence of peroxide to n-propyl chloride.
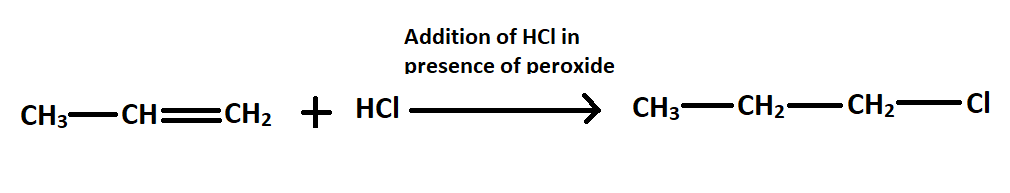
Note:
In case of haloalkanes containing more than three carbon atoms in the main chain are capable of having more than one $\beta $-positions. In such a case where more than one $\beta $-position is present, the hydrogen halide can be eliminated in more than one way. Thus one major and minor product is formed and the major product is decided by Saytzeff’s rule.
Complete answer:
-When haloalkanes having $\beta $-hydrogen atoms are heated in the presence of alcoholic solution of potassium hydroxide, then they undergo elimination reaction. Such reactions are also known as $\beta $- elimination reactions and results in the formation of alkenes.
-These reactions are known as $\beta $- elimination reactions because the hydrogen present at the $\beta $position of the alkanes is removed. The hydrogen atom which is attached to the carbon atom present next to that carbon atom which carries halogen is called $\beta $- hydrogen.
-To convert isopropyl chloride to n-propyl chloride first treat isopropyl chloride with alcoholic KOH to form prop$-1-$ene and eliminate HCl.

-When boiled with alcoholic KOH, the hydrogen atom transfers its electron pair to the adjacent carbon atom and the bromide gets removed. This forms a double bond between the $\alpha $ and $\beta $carbon atom.
- During the reaction halogen gets eliminated with hydrogen as hydrogen halide and thus alkene is formed.
-Then this HCl is treated with prop$-1-$ene in presence of peroxide to n-propyl chloride.

Note:
In case of haloalkanes containing more than three carbon atoms in the main chain are capable of having more than one $\beta $-positions. In such a case where more than one $\beta $-position is present, the hydrogen halide can be eliminated in more than one way. Thus one major and minor product is formed and the major product is decided by Saytzeff’s rule.
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Social Science: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

Why is the cell called the structural and functional class 12 biology CBSE

Draw ray diagrams each showing i myopic eye and ii class 12 physics CBSE

Which is the correct genotypic ratio of mendel dihybrid class 12 biology CBSE




