Borax, disodium tetraborate is an important boron compound, a mineral, and a salt of boric acid. The number of terminal B- - - - - OH presents in borax is?
(A) 3
(B) 4
(C) 5
(D) 6
Answer
582.9k+ views
Hint: Borax is also known as sodium borate, sodium tetraborate or disodium tetraborate. It is a colorless crystalline solid that dissolves in water. It has 2 four-coordinate boron centres and 2 three-coordinate boron centres.
Complete step by step solution:
In nature, borax combines to form boric acid and inorganic salts called borates. Borax is a form of hydrous sodium borate which occurs naturally in a region where evaporation of saline lake takes place.
Anhydrous sodium tetraborate or disodium tetraborate is represented by $ N{a_2}{B_4}{O_7} $ . From a chemical perspective borax has 2 four-coordinate boron centres and 2 three-coordinate boron centres.
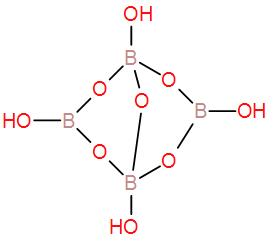
Disodium tetraborate has 4 B-OH bonds, as we can see in the structure.
Therefore, the correct answer is option B.
Note:
Borax is an important compound of boron because it has many important uses which are as follows, Borax is a component of many daily life products like detergents, cosmetics, and enamel glazes. Borax is used to make buffer solution, it is used as fire retardant, it is also used as an antifungal compound. Borax is used in the manufacture of fiberglass and as a flux in metallurgy. Borax is used as a neutron-capture shield for radioactive sources. It gives a good texture agent, therefore used in cooking. It is used in developing photographs. It is used as an alkali while developing photographs. It is also used as an insecticide. In artisanal gold mining, borax is used as a part of the process of eliminating the toxic mercury in the gold extraction process. Borax is used and added in flame to give it a yellow green color.
Borax is used for many cleaning purposes and can be used to make homemade laundry detergent. It even keeps ants and other pests away. It is also used as a flux in soldering and welding.
Borax even has a different number of molecules of water of crystallization such as sodium tetraborate pentahydrate and sodium tetraborate decahydrate.
Complete step by step solution:
In nature, borax combines to form boric acid and inorganic salts called borates. Borax is a form of hydrous sodium borate which occurs naturally in a region where evaporation of saline lake takes place.
Anhydrous sodium tetraborate or disodium tetraborate is represented by $ N{a_2}{B_4}{O_7} $ . From a chemical perspective borax has 2 four-coordinate boron centres and 2 three-coordinate boron centres.

Disodium tetraborate has 4 B-OH bonds, as we can see in the structure.
Therefore, the correct answer is option B.
Note:
Borax is an important compound of boron because it has many important uses which are as follows, Borax is a component of many daily life products like detergents, cosmetics, and enamel glazes. Borax is used to make buffer solution, it is used as fire retardant, it is also used as an antifungal compound. Borax is used in the manufacture of fiberglass and as a flux in metallurgy. Borax is used as a neutron-capture shield for radioactive sources. It gives a good texture agent, therefore used in cooking. It is used in developing photographs. It is used as an alkali while developing photographs. It is also used as an insecticide. In artisanal gold mining, borax is used as a part of the process of eliminating the toxic mercury in the gold extraction process. Borax is used and added in flame to give it a yellow green color.
Borax is used for many cleaning purposes and can be used to make homemade laundry detergent. It even keeps ants and other pests away. It is also used as a flux in soldering and welding.
Borax even has a different number of molecules of water of crystallization such as sodium tetraborate pentahydrate and sodium tetraborate decahydrate.
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Trending doubts
Which is more stable and why class 12 chemistry CBSE

Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

What are the major means of transport Explain each class 12 social science CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE




