Assertion: Diamond is the hardest substance on the earth.
Reason: It has a high melting point.
A. Both assertion and Reason are correct and reason is the correct explanation for Assertion.
B. Both assertion and Reason are correct and reason is not the correct explanation for Assertion.
C. Assertion is correct but Reason is incorrect.
D. Both Assertion and Reason are incorrect.
Answer
624k+ views
Hint: Hardness is the physical property of any substance which depends on many factors like the element it is made up of, the hybridization, the type of bond formed, the structure. There are different allotropic forms (chemically the same but vary in their physical properties) of carbon and diamond is one of those.
Complete answer:
Carbon belongs to the II period and IV group of the atomic table. It is therefore a metalloid which means its electronegativity and electropositivity is almost the same. As it is the first member of its group, carbon is the smallest among them and does not have d-orbitals. This element has the highest self-linking property among all the other elements in the periodic table. It is also able to form strong bonds with metals, nonmetals and metalloids. Diamond is a large molecule of carbon which does not have a specific formula.
Let us discuss the chemical and physical properties of diamond to find the correct explanation:
1. The hybridisation of carbon here is $\text{s}{{\text{p}}^{3}}$. Therefore it has a tetrahedral structure.
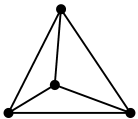
2. All four valencies of carbon atoms are filled in the structure of diamond. In diamond, the electrons are shared between four carbon atoms to form very strong chemical bonds forming an extremely rigid tetrahedral crystal. This tightly-bonded arrangement makes diamond as one of the hardest substances on Earth.
3. It is an insulator because there are no free electrons for the conduction of electricity.
4. The molecular arrangement of the carbon molecules are three dimensional in nature because of the tetrahedral geometry. This rarity in structure is formed only under massive pressure and high temperature. Hence, Diamond has a high melting point.
The correct answer to this question is option ‘b’, both assertion and Reason are correct and reason is not the correct explanation for Assertion.
So, the correct answer is “Option B”.
Note: Carbon has many allotropes but it does not mean that every allotrope will have the same physical and chemical properties. They are the same in many chemical properties which by the fact that they are composed of the same element than their macrostructure and other conditions that gave birth to their physical differences.
Complete answer:
Carbon belongs to the II period and IV group of the atomic table. It is therefore a metalloid which means its electronegativity and electropositivity is almost the same. As it is the first member of its group, carbon is the smallest among them and does not have d-orbitals. This element has the highest self-linking property among all the other elements in the periodic table. It is also able to form strong bonds with metals, nonmetals and metalloids. Diamond is a large molecule of carbon which does not have a specific formula.
Let us discuss the chemical and physical properties of diamond to find the correct explanation:
1. The hybridisation of carbon here is $\text{s}{{\text{p}}^{3}}$. Therefore it has a tetrahedral structure.

2. All four valencies of carbon atoms are filled in the structure of diamond. In diamond, the electrons are shared between four carbon atoms to form very strong chemical bonds forming an extremely rigid tetrahedral crystal. This tightly-bonded arrangement makes diamond as one of the hardest substances on Earth.
3. It is an insulator because there are no free electrons for the conduction of electricity.
4. The molecular arrangement of the carbon molecules are three dimensional in nature because of the tetrahedral geometry. This rarity in structure is formed only under massive pressure and high temperature. Hence, Diamond has a high melting point.
The correct answer to this question is option ‘b’, both assertion and Reason are correct and reason is not the correct explanation for Assertion.
So, the correct answer is “Option B”.
Note: Carbon has many allotropes but it does not mean that every allotrope will have the same physical and chemical properties. They are the same in many chemical properties which by the fact that they are composed of the same element than their macrostructure and other conditions that gave birth to their physical differences.
Recently Updated Pages
Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 English: Engaging Questions & Answers for Success

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 11 Social Science: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

Difference Between Prokaryotic Cells and Eukaryotic Cells

Two of the body parts which do not appear in MRI are class 11 biology CBSE

1 ton equals to A 100 kg B 1000 kg C 10 kg D 10000 class 11 physics CBSE

Draw a diagram of nephron and explain its structur class 11 biology CBSE

10 examples of friction in our daily life




