What are the structures of metaphosphoric acid and trimetaphosphoric acid?
Answer
626.7k+ views
Hint: Metaphosphoric acid and trimetaphosphoric acid are the oxoacids of phosphorus. Phosphorus forms a double bond with the O atom and forms a single bond with the OH group.
Complete answer:
Metaphosphoric acid: It is the oxoacid of phosphorus which has a formula \[HP{{O}_{3}}\].
Metaphosphoric acid has three forms: i) single-molecule form, ii) trimetaphosphoric acid which has a cyclo structure, iii) poly metaphosphoric acid which has a linear polymer chain structure.
In metaphosphoric acid, the phosphorus is in the +5 oxidation state.
In the structure, it forms two double bonds with the oxygen atom and one single bond with the OH group.
The structure of metaphosphoric acid is given below:
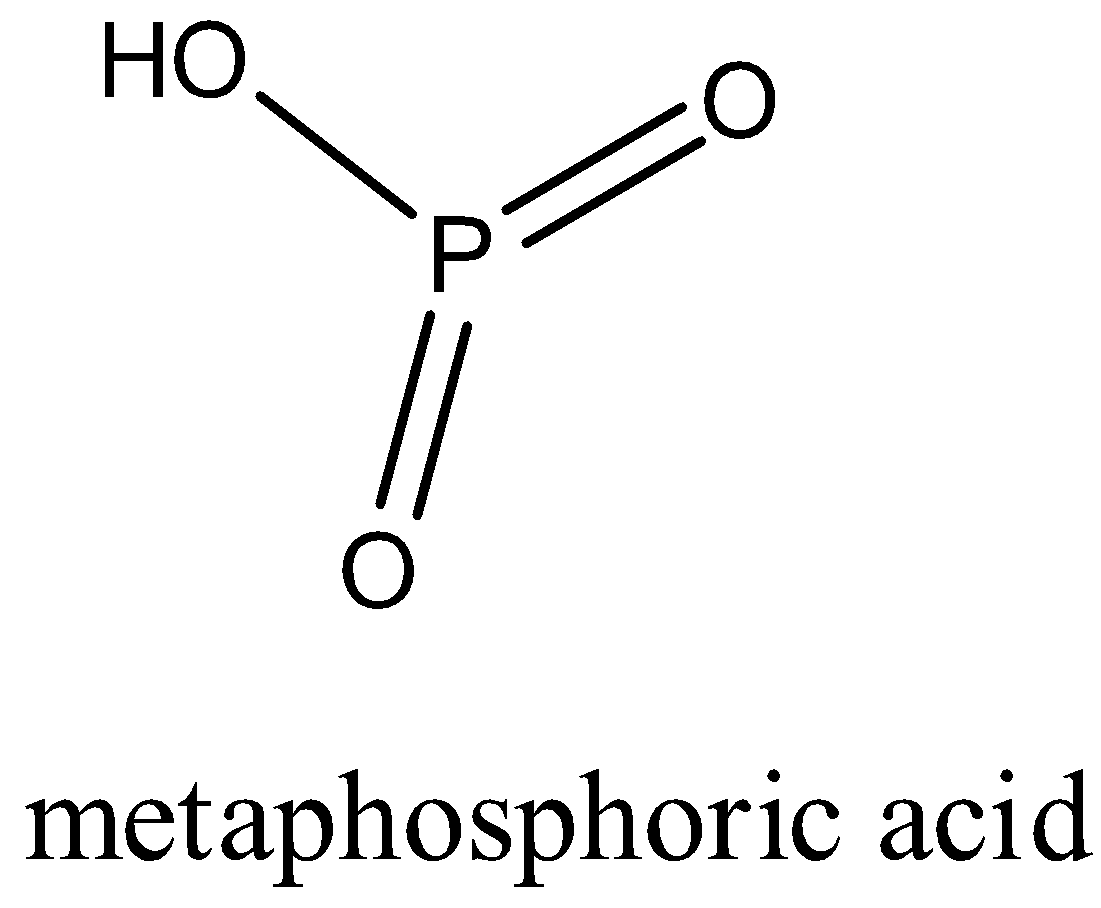
It is a monobasic compound.
Trimetaphosphoric acid: It is also an oxoacid of phosphorus which has a formula \[{{(HP{{O}_{3}})}_{3}}\].
It is commonly called cyclotrimetaphosphoric acid because they form a cyclic structure. The three metaphosphoric acid molecules form a ring structure.
In trimetaphosphoric acid, the phosphorus is in the +5 oxidation state.
In this structure, there are three P-OH bonds, three P=O bonds, and three P-O-P bonds.
The structure of trimetaphosphoric acid is given below:
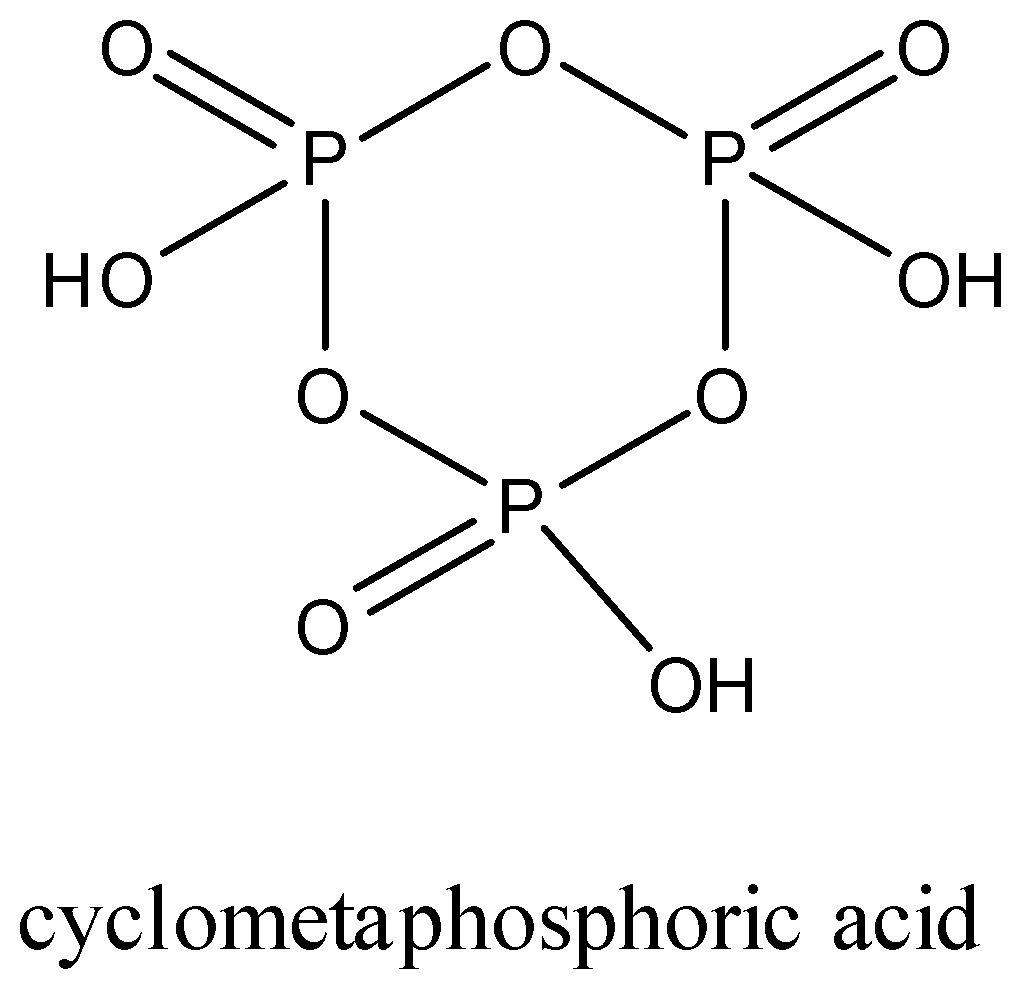
It is a tribasic compound.
Additional Information:
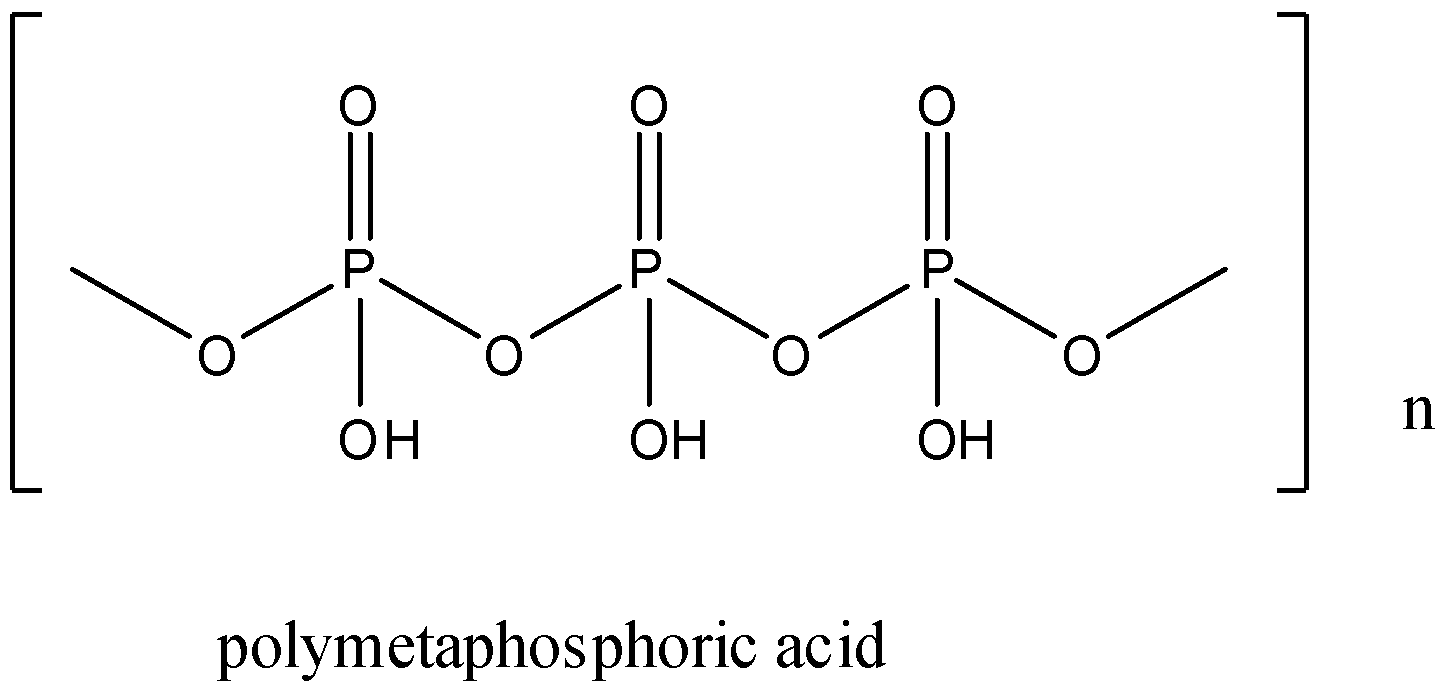
There is one more form of metaphosphoric acid, which is called poly metaphosphoric acid. The formula of poly metaphosphoric acid is \[{{(HP{{O}_{3}})}_{n}}\].
It has a linear polymer structure. The oxidation state of phosphorus is +5.
The structure of poly metaphosphoric acid is given below:
Note: According to the structure of the oxoacids of phosphorus we can find the basicity of the compound. The number of OH groups in the compound is equal to the basicity of the compound.
Complete answer:
Metaphosphoric acid: It is the oxoacid of phosphorus which has a formula \[HP{{O}_{3}}\].
Metaphosphoric acid has three forms: i) single-molecule form, ii) trimetaphosphoric acid which has a cyclo structure, iii) poly metaphosphoric acid which has a linear polymer chain structure.
In metaphosphoric acid, the phosphorus is in the +5 oxidation state.
In the structure, it forms two double bonds with the oxygen atom and one single bond with the OH group.
The structure of metaphosphoric acid is given below:

It is a monobasic compound.
Trimetaphosphoric acid: It is also an oxoacid of phosphorus which has a formula \[{{(HP{{O}_{3}})}_{3}}\].
It is commonly called cyclotrimetaphosphoric acid because they form a cyclic structure. The three metaphosphoric acid molecules form a ring structure.
In trimetaphosphoric acid, the phosphorus is in the +5 oxidation state.
In this structure, there are three P-OH bonds, three P=O bonds, and three P-O-P bonds.
The structure of trimetaphosphoric acid is given below:

It is a tribasic compound.
Additional Information:
There is one more form of metaphosphoric acid, which is called poly metaphosphoric acid. The formula of poly metaphosphoric acid is \[{{(HP{{O}_{3}})}_{n}}\].
It has a linear polymer structure. The oxidation state of phosphorus is +5.
The structure of poly metaphosphoric acid is given below:

Note: According to the structure of the oxoacids of phosphorus we can find the basicity of the compound. The number of OH groups in the compound is equal to the basicity of the compound.
Recently Updated Pages
Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 English: Engaging Questions & Answers for Success

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 9 General Knowledge: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

Why is the cell called the structural and functional class 12 biology CBSE

Draw ray diagrams each showing i myopic eye and ii class 12 physics CBSE

Which is the correct genotypic ratio of mendel dihybrid class 12 biology CBSE




