What are the possible isomers for ${C_3}{H_7}NO$?
Answer
549k+ views
Hint: We can say that a molecule could contain a specific shape in space which could contribute to its nature. We have to know that isomers are molecules that are an indistinguishable number of atoms of each element yet several arrangements of atoms in space. Isomerism is the presence or plausibility of isomers.
Complete step by step answer:
We have to know that structural isomers of the compounds exhibit the same molecular formula but vary in the spatial arrangement of atoms and the state is called structural isomerism. Based on IUPAC naming, we could call structural isomerism as constitutional isomerism.
We can group structural isomers into three kinds,
- Chain isomers: The atoms of carbon are arranged in several sequences.
- Position isomers: The skeletal carbon chain remains unchanged, but the location of the functional group is varied. This classification of structural isomers is known as position isomers and the isomerism is called positional isomerism.
- Example: Compounds that have a molecular formula ${C_3}{H_7}Br$ would be 1-bromopropane and 2-bromopropane. We can draw as,
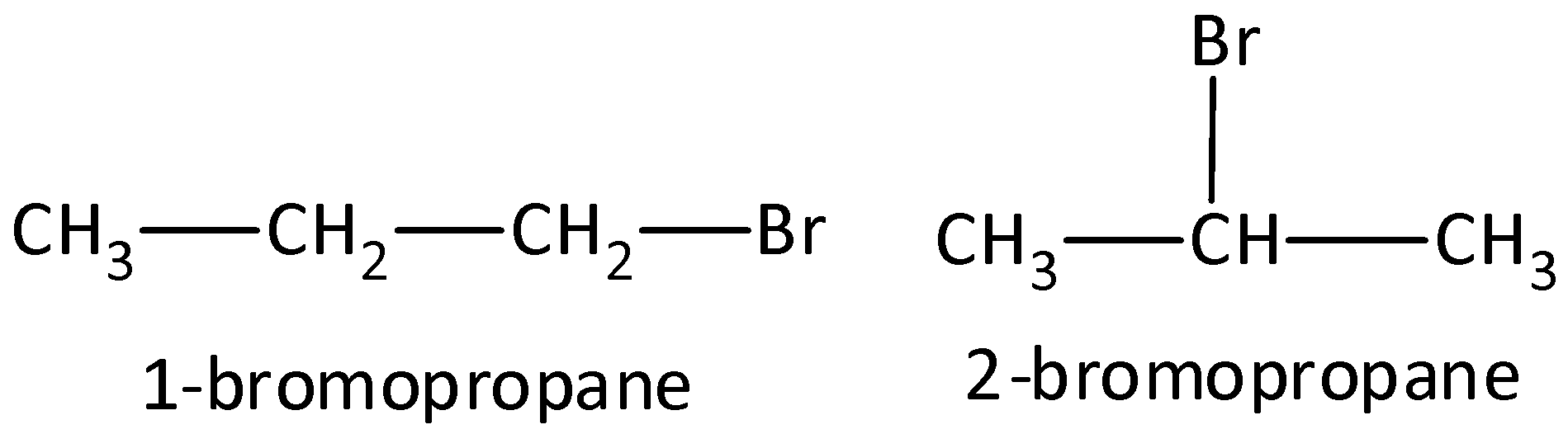
- Functional group isomers: The arrangement of atoms to make several various functional groups is known as functional group isomers and this is known as functional group isomerism. Example: Compounds that have a molecular formula ${C_3}{H_6}O$ would be propanal (aldehyde) or a propanone (ketone). We can draw the structure as,
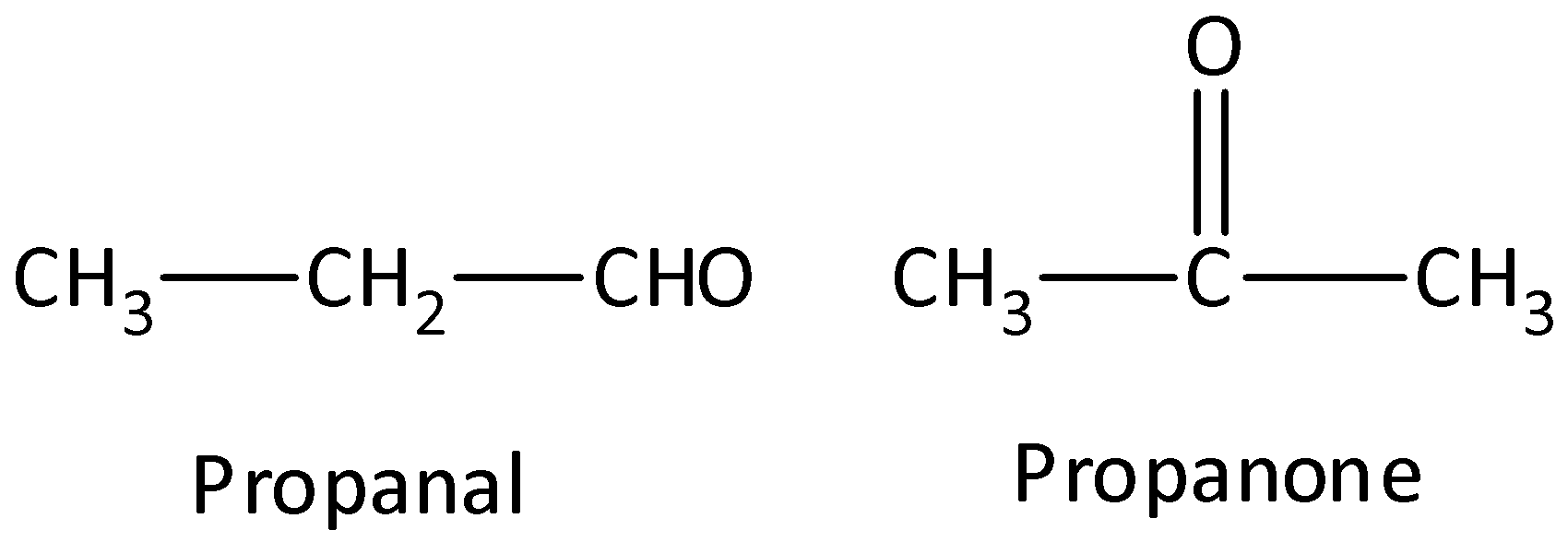
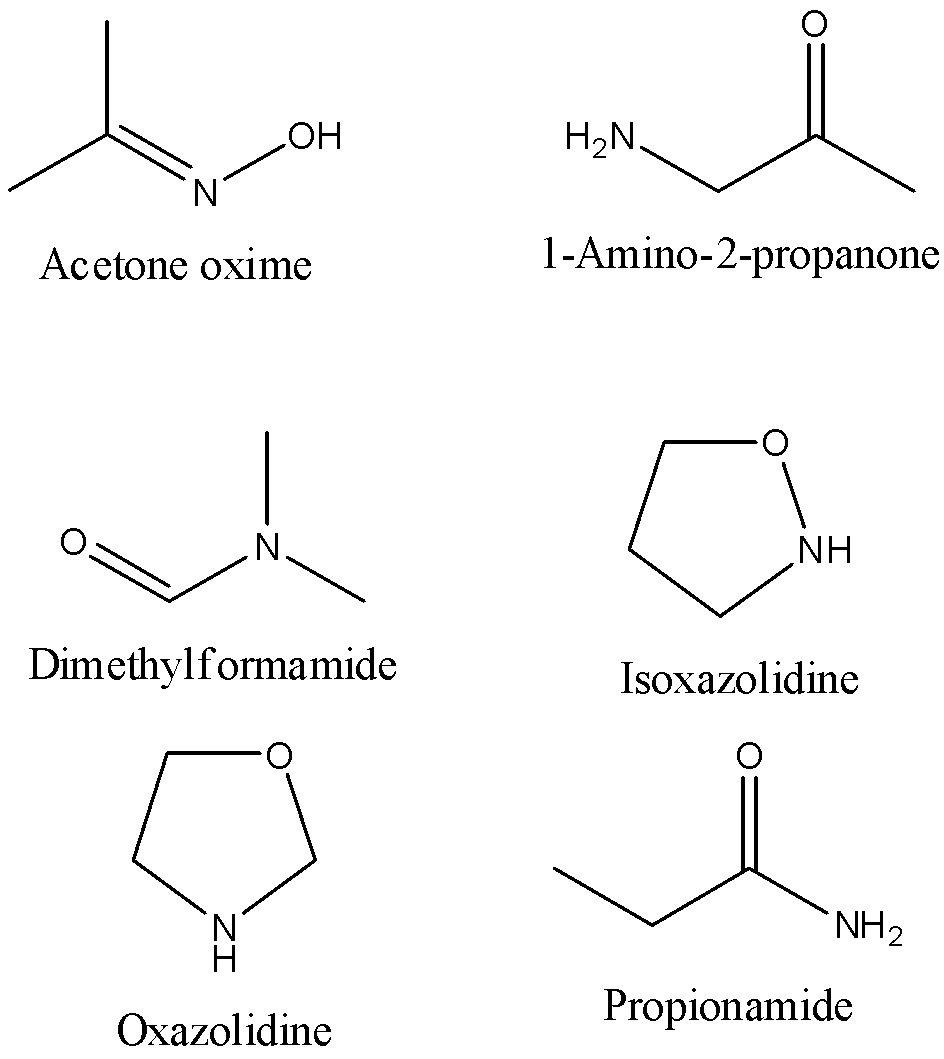
The possible isomers of ${C_3}{H_7}NO$ are,
- Acetone oxime
- 1-Amino-2-propanone
- Dimethylformamide
- Isoxazolidine
- Oxazolidine
- Propionamide
We can draw the structure of these isomers as,
Note: We can say that structural isomers exhibit different chemical properties like lower melting and boiling points etc. We can say examples of stereoisomers as cis 2-butene and trans 2-butene. We can say examples of chain isomers as n-pentane, isopentane and neopentane.
Complete step by step answer:
We have to know that structural isomers of the compounds exhibit the same molecular formula but vary in the spatial arrangement of atoms and the state is called structural isomerism. Based on IUPAC naming, we could call structural isomerism as constitutional isomerism.
We can group structural isomers into three kinds,
- Chain isomers: The atoms of carbon are arranged in several sequences.
- Position isomers: The skeletal carbon chain remains unchanged, but the location of the functional group is varied. This classification of structural isomers is known as position isomers and the isomerism is called positional isomerism.
- Example: Compounds that have a molecular formula ${C_3}{H_7}Br$ would be 1-bromopropane and 2-bromopropane. We can draw as,
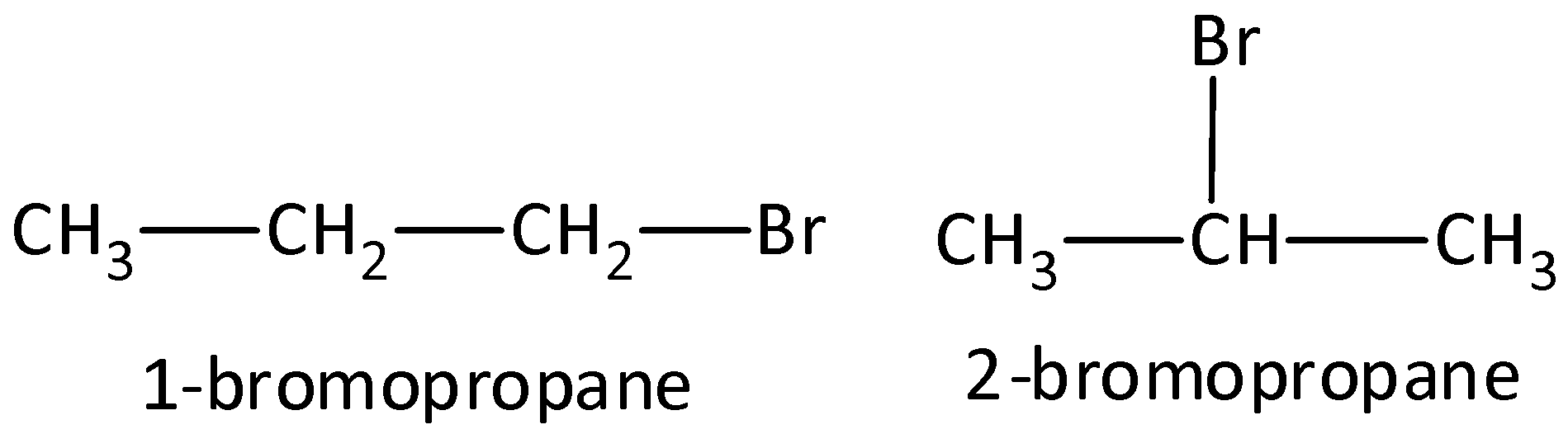
- Functional group isomers: The arrangement of atoms to make several various functional groups is known as functional group isomers and this is known as functional group isomerism. Example: Compounds that have a molecular formula ${C_3}{H_6}O$ would be propanal (aldehyde) or a propanone (ketone). We can draw the structure as,

The possible isomers of ${C_3}{H_7}NO$ are,
- Acetone oxime
- 1-Amino-2-propanone
- Dimethylformamide
- Isoxazolidine
- Oxazolidine
- Propionamide
We can draw the structure of these isomers as,

Note: We can say that structural isomers exhibit different chemical properties like lower melting and boiling points etc. We can say examples of stereoisomers as cis 2-butene and trans 2-butene. We can say examples of chain isomers as n-pentane, isopentane and neopentane.
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Trending doubts
Which is more stable and why class 12 chemistry CBSE

Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

What are the major means of transport Explain each class 12 social science CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE




