What are Peptides and peptide linkage? Explain with an example.
Answer
615k+ views
Hint: Peptides have a very important role. In our body there are a total of amino acids present. These amino acids are further categorised as essential and non-essential amino acids.
Complete step by step solution
Amino acids are a special kind of organic compound which contains both amino and carboxyl groups. These amino acids are joined together to form proteins and we all know how important proteins are in our daily life.
These amino acids are joined by a condensation reaction. In this condensation reaction, amino acids of one amino acid will reacts with carboxyl group of another amino acids. And water will be eliminated as a by-product’s product of this reaction is peptide. And the bond formed between amino acids is known as peptide bond which is actually an amide bond.
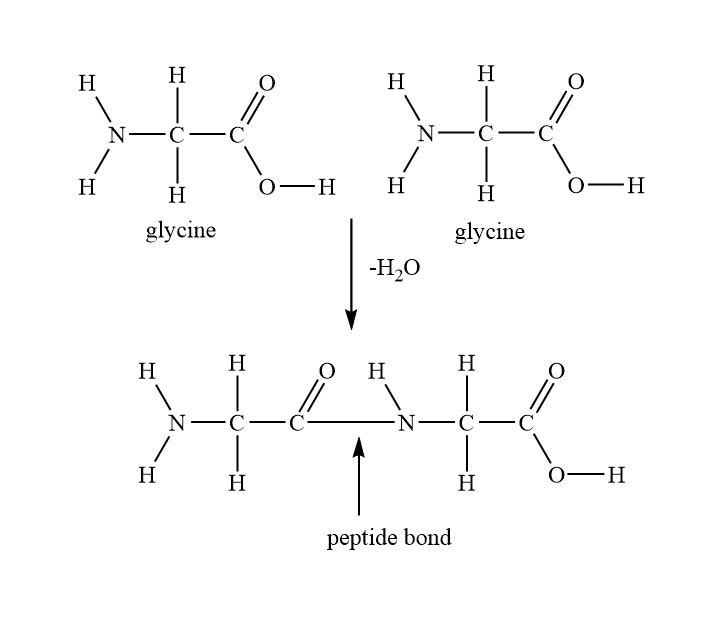
And the naming of these peptides will depend on the number of amino acids involved in the reaction. For example, if two amino acids will involve then the peptide bond is known as dipeptide.
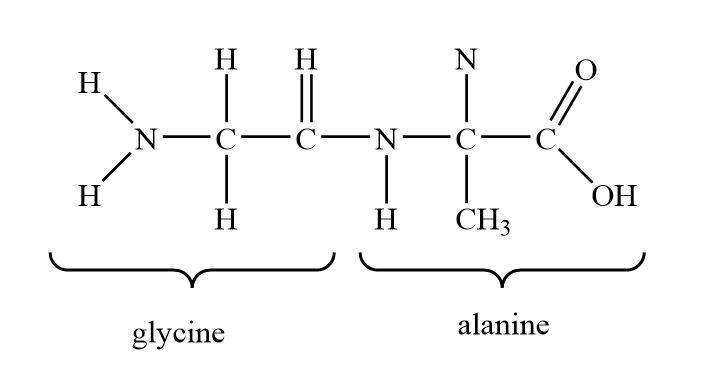
If the peptide is formed by three amino acids then the peptide form will be known as tripeptides.
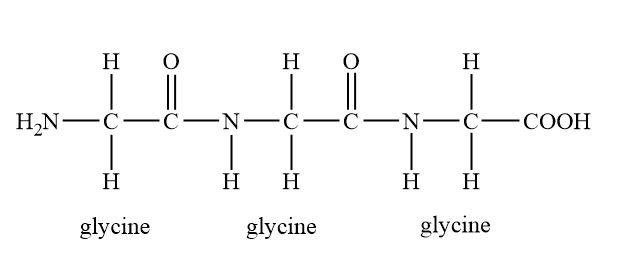
And if amino acids greater than fifty amino will involve then the peptide form is called polypeptides.
Note:
There is a unique way of naming in the peptides. The naming of peptides depends upon the part of the amino acids that are involved in the condensation reaction. The naming is done in a way that it should be N-terminal (amino terminal) to C-terminal (carboxylic terminal).
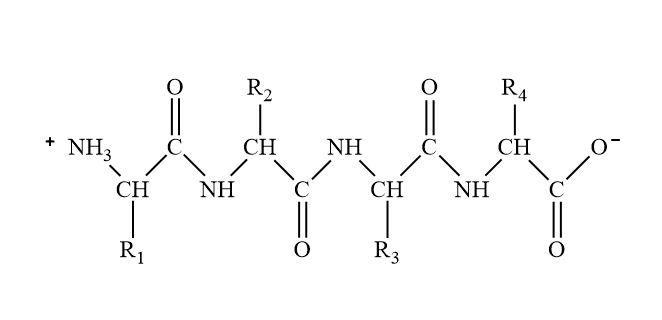
Complete step by step solution
Amino acids are a special kind of organic compound which contains both amino and carboxyl groups. These amino acids are joined together to form proteins and we all know how important proteins are in our daily life.
These amino acids are joined by a condensation reaction. In this condensation reaction, amino acids of one amino acid will reacts with carboxyl group of another amino acids. And water will be eliminated as a by-product’s product of this reaction is peptide. And the bond formed between amino acids is known as peptide bond which is actually an amide bond.

And the naming of these peptides will depend on the number of amino acids involved in the reaction. For example, if two amino acids will involve then the peptide bond is known as dipeptide.

If the peptide is formed by three amino acids then the peptide form will be known as tripeptides.

And if amino acids greater than fifty amino will involve then the peptide form is called polypeptides.
Note:
There is a unique way of naming in the peptides. The naming of peptides depends upon the part of the amino acids that are involved in the condensation reaction. The naming is done in a way that it should be N-terminal (amino terminal) to C-terminal (carboxylic terminal).

Recently Updated Pages
Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 English: Engaging Questions & Answers for Success

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 9 General Knowledge: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

Why is the cell called the structural and functional class 12 biology CBSE

Draw ray diagrams each showing i myopic eye and ii class 12 physics CBSE

Which is the correct genotypic ratio of mendel dihybrid class 12 biology CBSE




