What are antioxidants? Give two examples.
Answer
600k+ views
Hint: Think about what antioxidants are. We see a lot of advertisements on television. Generally the ads pertaining to butter and edible oils like sunflower oil or rice bran oil, etc. say the oil has antioxidant properties. Think about what happens to oils after a long period of time and what the chemistry behind it is.
Complete answer:
- Antioxidants are substances which when added to food, retards or prevents the oxidative deterioration of food.
- Fats and oils are oxidized easily and they become rancid and unpalatable.
- Addition of antioxidants prevents their oxidation and prevents them from becoming rancid.
- When food gets oxidized, free radicals are generated which have bad effects on our body. Antioxidants react with these free radicals and stop further oxidation of food.
- Examples of antioxidants are Butylated hydroxytoluene, BHT and Butylated hydroxyanisole, BHA.
- Butylated hydroxytoluene, BHT has the molecular formula, ${{C}_{15}}{{H}_{24}}O$. The free radical formed due to oxidation of fat and oil reacts with BHT to form BHT radical which is stable due to electron clouds of the benzene ring. BHT radical doesn’t react further and thus, stops the chain reaction and prevents oxidative rancidity of fats. It used to retain food odour, colour and flavor.
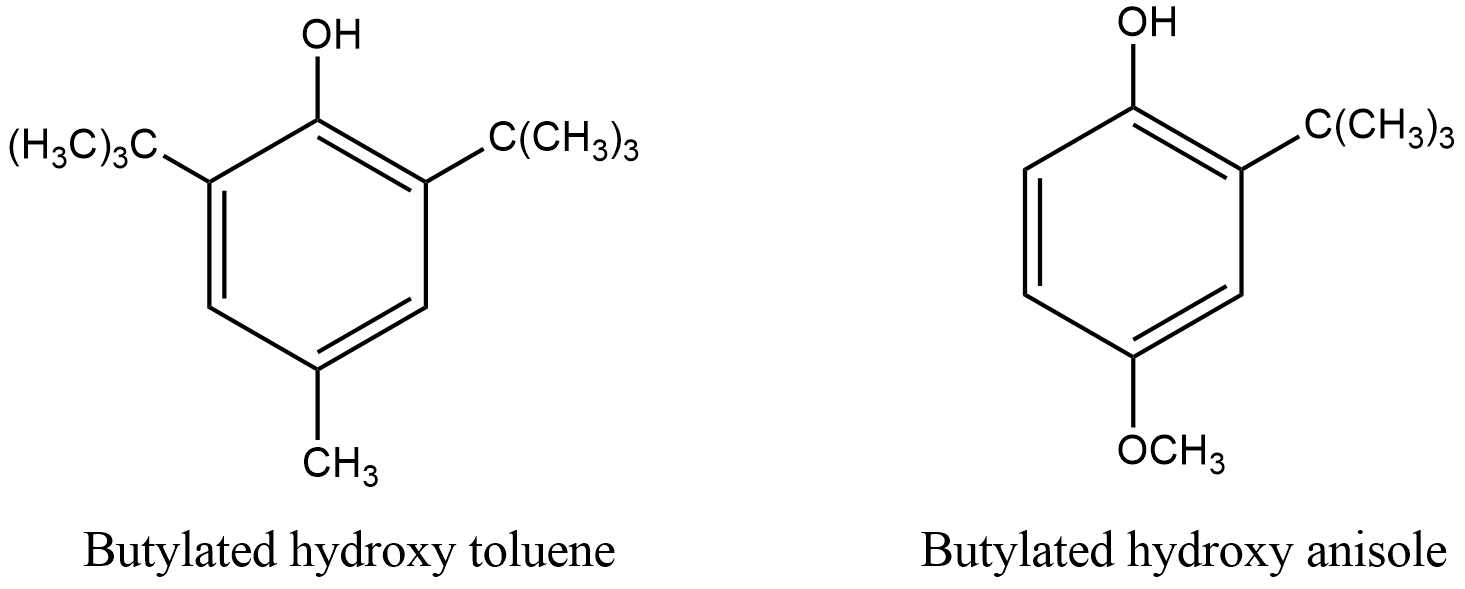
- Butylated hydroxyanisole, BHA has the molecular formula, ${{C}_{11}}{{H}_{16}}{{O}_{2}}$. BHA is generally used to preserve fats and prevent them from becoming rancid and is similar to BHT. BHA is found in butter, meat, cereals, chewing gums, snack foods, etc. It is also found in rubber products, cosmetics and petroleum products.
Note:
Remember fats and oils get easily oxidized and turn rancid after a period of time. In order to avoid oxidation, antioxidants are used. Antioxidants are the chemicals which prevent oxidation of substances by reacting with the free radicals generated during oxidation of foods.
Complete answer:
- Antioxidants are substances which when added to food, retards or prevents the oxidative deterioration of food.
- Fats and oils are oxidized easily and they become rancid and unpalatable.
- Addition of antioxidants prevents their oxidation and prevents them from becoming rancid.
- When food gets oxidized, free radicals are generated which have bad effects on our body. Antioxidants react with these free radicals and stop further oxidation of food.
- Examples of antioxidants are Butylated hydroxytoluene, BHT and Butylated hydroxyanisole, BHA.
- Butylated hydroxytoluene, BHT has the molecular formula, ${{C}_{15}}{{H}_{24}}O$. The free radical formed due to oxidation of fat and oil reacts with BHT to form BHT radical which is stable due to electron clouds of the benzene ring. BHT radical doesn’t react further and thus, stops the chain reaction and prevents oxidative rancidity of fats. It used to retain food odour, colour and flavor.

- Butylated hydroxyanisole, BHA has the molecular formula, ${{C}_{11}}{{H}_{16}}{{O}_{2}}$. BHA is generally used to preserve fats and prevent them from becoming rancid and is similar to BHT. BHA is found in butter, meat, cereals, chewing gums, snack foods, etc. It is also found in rubber products, cosmetics and petroleum products.
Note:
Remember fats and oils get easily oxidized and turn rancid after a period of time. In order to avoid oxidation, antioxidants are used. Antioxidants are the chemicals which prevent oxidation of substances by reacting with the free radicals generated during oxidation of foods.
Recently Updated Pages
Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 English: Engaging Questions & Answers for Success

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 9 General Knowledge: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

Why is the cell called the structural and functional class 12 biology CBSE

Draw ray diagrams each showing i myopic eye and ii class 12 physics CBSE

Which is the correct genotypic ratio of mendel dihybrid class 12 biology CBSE




