An organic compound with the molecular formula \[{{\text{C}}_{\text{9}}}{{\text{H}}_{{\text{10}}}}{\text{O}}\] forms 2,4-DNP derivative, reduces Tollens reagent and undergoes Cannizzaro reaction. On vigorous oxidation, it gives 1,2-benzenedicarboxylic acid. Identify the compound.
Answer
606.9k+ views
Hint: From the given molecular formula, calculate the degree of unsaturation. You can confirm the presence of the carbonyl group by the reaction with 2,4-DNP. You can use the test with Tollen’s reagent to confirm presence of an aldehyde group. From the product obtained on vigorous oxidation, you can determine the structure of the starting material.
Complete step by step answer:
The organic compound has the molecular formula \[{{\text{C}}_{\text{9}}}{{\text{H}}_{{\text{10}}}}{\text{O}}\]
The molecular formula of the corresponding saturated compound is \[{{\text{C}}_{\text{9}}}{{\text{H}}_{{\text{20}}}}{\text{O}}\] .
Calculate the degree of unsaturation:
\[\dfrac{{20 - 10}}{2} = \dfrac{{10}}{2} = 5\]
A benzene nucleus has a degree of unsaturation of 4. A benzene nucleus has one ring and three carbon-carbon double bonds.
Thus, the given compound with degree of unsaturation of 5 has a benzene nucleus and a double bond.
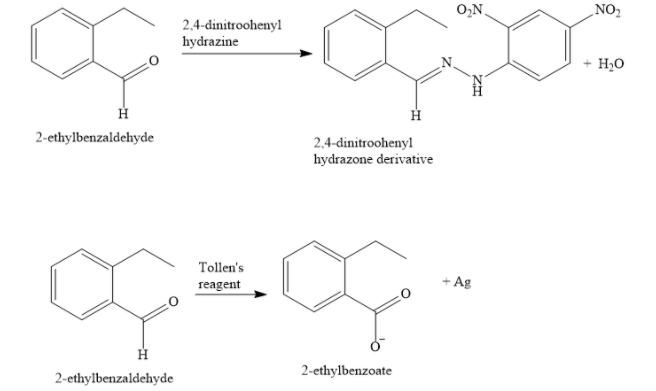
The given compound forms 2,4-DNP derivative and also reduces Tollens reagent.
From this information, you can conclude that the compound is an aldehyde. An aldehyde contains the \[ - {\text{CHO}}\] group.
The compound undergoes Cannizzaro reaction. Hence, an alpha H atom is absent in this compound.
During the Cannizzaro reaction, one molecule of aldehyde is oxidized to carboxylate ion and the other molecule is reduced to alcohol. Those aldehydes contain an alpha hydrogen atom, they do not undergo Cannizzaro reaction. Instead, they undergo an aldol condensation reaction.
When you subject the given organic compound to vigorous oxidation, you obtain 1,2-benzenedicarboxylic acid. Hence, you can conclude that the aldehyde group is directly linked to the benzene nucleus. You can also say that the compound is ortho di-substituted benzene.
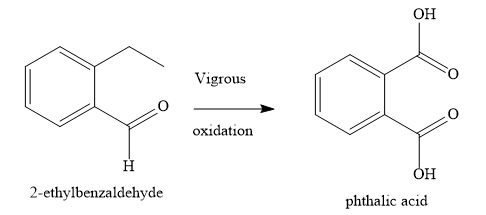
Hence, the given compound.is 2-ethylbenzaldehyde.
Note: An aldehyde (or a ketone) reacts with 2,4-DNP to form 2,4-dinitrophenyl hydrazone derivative. Here, 2,4-DNP reagent means 2,4-dinitrophenyl hydrazine. Aldehydes are oxidized by Tollen’s reagent to give black silver mirror. Tollen’s reagent is an ammoniacal silver nitrate solution. Ketones do not react with Tollen’s reagent.
Silver mirror test is used to distinguish between aldehydes and ketones.
Complete step by step answer:
The organic compound has the molecular formula \[{{\text{C}}_{\text{9}}}{{\text{H}}_{{\text{10}}}}{\text{O}}\]
The molecular formula of the corresponding saturated compound is \[{{\text{C}}_{\text{9}}}{{\text{H}}_{{\text{20}}}}{\text{O}}\] .
Calculate the degree of unsaturation:
\[\dfrac{{20 - 10}}{2} = \dfrac{{10}}{2} = 5\]
A benzene nucleus has a degree of unsaturation of 4. A benzene nucleus has one ring and three carbon-carbon double bonds.
Thus, the given compound with degree of unsaturation of 5 has a benzene nucleus and a double bond.
The given compound forms 2,4-DNP derivative and also reduces Tollens reagent.
From this information, you can conclude that the compound is an aldehyde. An aldehyde contains the \[ - {\text{CHO}}\] group.
The compound undergoes Cannizzaro reaction. Hence, an alpha H atom is absent in this compound.
During the Cannizzaro reaction, one molecule of aldehyde is oxidized to carboxylate ion and the other molecule is reduced to alcohol. Those aldehydes contain an alpha hydrogen atom, they do not undergo Cannizzaro reaction. Instead, they undergo an aldol condensation reaction.
When you subject the given organic compound to vigorous oxidation, you obtain 1,2-benzenedicarboxylic acid. Hence, you can conclude that the aldehyde group is directly linked to the benzene nucleus. You can also say that the compound is ortho di-substituted benzene.


Hence, the given compound.is 2-ethylbenzaldehyde.
Note: An aldehyde (or a ketone) reacts with 2,4-DNP to form 2,4-dinitrophenyl hydrazone derivative. Here, 2,4-DNP reagent means 2,4-dinitrophenyl hydrazine. Aldehydes are oxidized by Tollen’s reagent to give black silver mirror. Tollen’s reagent is an ammoniacal silver nitrate solution. Ketones do not react with Tollen’s reagent.
Silver mirror test is used to distinguish between aldehydes and ketones.
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Name the crygenes that control cotton bollworm and class 12 biology CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

Ribosomal RNA is actively synthesised in A Nucleoplasm class 12 biology CBSE

How many molecules of ATP and NADPH are required information class 12 biology CBSE




