An organic compound ${{C}_{3}}{{H}_{9}}N$ (A) when treated with nitrous acid, gave an alcohol and ${{N}_{2}}$ gas was evolved. (A) on warming with $CHC{{l}_{3}}$ and caustic potash gave (C) which on reduction gave isopropylmethylamine. Predict the structure of (A).
[A] $C{{H}_{3}}C{{H}_{2}}NHC{{H}_{3}}$
[B] $C{{H}_{3}}NC{{H}_{3}}C{{H}_{3}}$
[C] $C{{H}_{3}}C{{H}_{2}}C{{H}_{2}}N{{H}_{2}}$
[D] ${{\left( C{{H}_{3}} \right)}_{2}}CHN{{H}_{2}}$
Answer
604.2k+ views
Hint: To solve this, remember that primary amine react with acids to give alcohol. Identify the primary amines from the given option. Remember that the product obtained is isopropylmethylamine. So, the starting product must be an iso-amine and not n-amine.
Complete answer:
Here first of all we can see that (A) upon reaction with an acid gives an alcohol. We know that only a primary amine upon reaction with an acid gives us alcohol.
So, we can surely say that A is a primary amine. We know that primary amines are the amines having only one carbon attached to the nitrogen atom.
Also, it is given that the amine A on warming with chloroform and potassium hydroxide reduces to isopropylmethylamine.
We know that the structure of isopropylmethylamine is ${{\left( C{{H}_{3}} \right)}_{2}}CHNHC{{H}_{3}}$
Now let us see the given options and find out the primary amine among them.
In the first option we have $C{{H}_{3}}C{{H}_{2}}NHC{{H}_{3}}$ which is a secondary amine.
Then we have $C{{H}_{3}}NC{{H}_{3}}C{{H}_{3}}$ which is a tertiary amine.
Next we have $C{{H}_{3}}C{{H}_{2}}C{{H}_{2}}N{{H}_{2}}$ which is a primary amine and the last option ${{\left( C{{H}_{3}} \right)}_{2}}CHN{{H}_{2}}$ is also a primary amine.
Among these two, $C{{H}_{3}}C{{H}_{2}}C{{H}_{2}}N{{H}_{2}}$ is propylamine and ${{\left( C{{H}_{3}} \right)}_{2}}CHN{{H}_{2}}$ is isopropyl amine. So, either of them can be the correct answer.
But upon reduction, we obtain isopropylmethylamine so the correct answer should be the isopropyl amine and not n-propylamine.
We can write the reaction as-
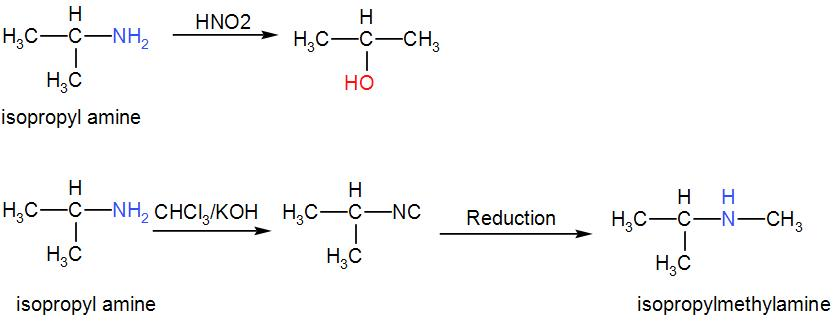
We can understand from the above discussion that the correct answer is option [D]${{\left( C{{H}_{3}} \right)}_{2}}CHN{{H}_{2}}$.
Note:
Just like we’ve seen in the above discussion that primary amines give us alcohol with acid, similarly it also gives us a diazonium salt which forms alcohols on addition of water but secondary amines have one hydrogen atom attached on them therefore, they cannot complete the diazotization reaction and give us a yellow oily nitrosamine product. Three degree amines have no hydrogen atoms attached to them thus they undergo simple acid- base reactions and give us soluble salts.
Complete answer:
Here first of all we can see that (A) upon reaction with an acid gives an alcohol. We know that only a primary amine upon reaction with an acid gives us alcohol.
So, we can surely say that A is a primary amine. We know that primary amines are the amines having only one carbon attached to the nitrogen atom.
Also, it is given that the amine A on warming with chloroform and potassium hydroxide reduces to isopropylmethylamine.
We know that the structure of isopropylmethylamine is ${{\left( C{{H}_{3}} \right)}_{2}}CHNHC{{H}_{3}}$
Now let us see the given options and find out the primary amine among them.
In the first option we have $C{{H}_{3}}C{{H}_{2}}NHC{{H}_{3}}$ which is a secondary amine.
Then we have $C{{H}_{3}}NC{{H}_{3}}C{{H}_{3}}$ which is a tertiary amine.
Next we have $C{{H}_{3}}C{{H}_{2}}C{{H}_{2}}N{{H}_{2}}$ which is a primary amine and the last option ${{\left( C{{H}_{3}} \right)}_{2}}CHN{{H}_{2}}$ is also a primary amine.
Among these two, $C{{H}_{3}}C{{H}_{2}}C{{H}_{2}}N{{H}_{2}}$ is propylamine and ${{\left( C{{H}_{3}} \right)}_{2}}CHN{{H}_{2}}$ is isopropyl amine. So, either of them can be the correct answer.
But upon reduction, we obtain isopropylmethylamine so the correct answer should be the isopropyl amine and not n-propylamine.
We can write the reaction as-

We can understand from the above discussion that the correct answer is option [D]${{\left( C{{H}_{3}} \right)}_{2}}CHN{{H}_{2}}$.
Note:
Just like we’ve seen in the above discussion that primary amines give us alcohol with acid, similarly it also gives us a diazonium salt which forms alcohols on addition of water but secondary amines have one hydrogen atom attached on them therefore, they cannot complete the diazotization reaction and give us a yellow oily nitrosamine product. Three degree amines have no hydrogen atoms attached to them thus they undergo simple acid- base reactions and give us soluble salts.
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Trending doubts
Which is more stable and why class 12 chemistry CBSE

Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

What are the major means of transport Explain each class 12 social science CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE




