An organic compound (A) with molecular formula, \[{{\text{C}}_{\text{8}}}{{\text{H}}_{\text{8}}}{\text{O}}\] forms an orange-red precipitate with 2,4-DNP reagent and gives yellow precipitate on heating with iodine in the presence of sodium hydroxide. lt neither reduces Tollens' or Fehling's reagent, nor does it decolourise bromine water or Baeyer's reagent. On drastic oxidation with chromic acid, it gives a carboxylic acid (B) having molecular formula, \[{{\text{C}}_{\text{7}}}{{\text{H}}_{\text{6}}}{{\text{O}}_2}\] . Identify the compounds (A) and (B) and explain the reactions involved.
Answer
583.5k+ views
Hint:From the given molecular formula, calculate the degree of unsaturation. You can confirm the presence of the carbonyl group by the reaction with 2,4-DNP. You can use the test with Tollen’s reagent or with Fehling’s solution to confirm absence of an aldehyde group. From the product obtained on vigorous oxidation, you can determine the structure of the starting material.
Complete answer:
The organic compound has the molecular formula \[{{\text{C}}_{\text{8}}}{{\text{H}}_{\text{8}}}{\text{O}}\]
The molecular formula of the corresponding saturated compound is \[{{\text{C}}_{\text{8}}}{{\text{H}}_{{\text{18}}}}{\text{O}}\] .
Calculate the degree of unsaturation:
\[\dfrac{{18 - 8}}{2} = \dfrac{{10}}{2} = 5\]
A benzene nucleus has a degree of unsaturation of 4. A benzene nucleus has one ring and three carbon-carbon double bonds.
Thus, the given compound with degree of unsaturation of 5 has a benzene nucleus and a double bond.
The compound (A) forms an orange-red precipitate with 2,4-DNP reagent. This confirms the presence of the carbonyl group. The compound (A) gives yellow precipitate on heating with iodine in the presence of sodium hydroxide. This confirms the presence of methyl keto groups.
The compound (A) neither reduces Tollens' or Fehling's reagent. It confirms the absence of the aldehyde group.
The compound (A) neither decolourises bromine water nor Baeyer's reagent. It confirms the absence of carbon-carbon double or triple bonds.
From this information, you can conclude that the compound is acetophenone.
When you subject acetophenone to vigorous oxidation, you obtain benzoic acid (compound (B) with molecular formula \[{{\text{C}}_{\text{7}}}{{\text{H}}_{\text{6}}}{{\text{O}}_2}\] .
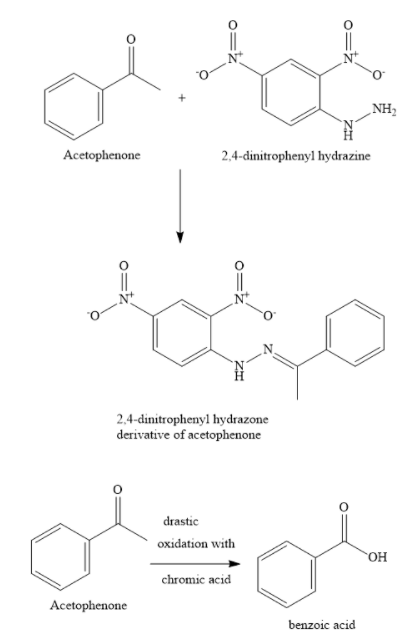
Hence, the compound (A) is acetophenone and the compound (B) is benzoic acid.
Note:
An aldehyde (or a ketone) reacts with 2,4-DNP to form a 2,4-dinitrophenyl hydrazone derivative. Here, 2,4-DNP reagent means 2,4-dinitrophenyl hydrazine. Aldehydes are oxidized by Tollen’s reagent to give black silver mirror. Tollen’s reagent is an ammoniacal silver nitrate solution. Ketones do not react with Tollen’s reagent. Silver mirror test is used to distinguish between aldehydes and ketones.
Complete answer:
The organic compound has the molecular formula \[{{\text{C}}_{\text{8}}}{{\text{H}}_{\text{8}}}{\text{O}}\]
The molecular formula of the corresponding saturated compound is \[{{\text{C}}_{\text{8}}}{{\text{H}}_{{\text{18}}}}{\text{O}}\] .
Calculate the degree of unsaturation:
\[\dfrac{{18 - 8}}{2} = \dfrac{{10}}{2} = 5\]
A benzene nucleus has a degree of unsaturation of 4. A benzene nucleus has one ring and three carbon-carbon double bonds.
Thus, the given compound with degree of unsaturation of 5 has a benzene nucleus and a double bond.
The compound (A) forms an orange-red precipitate with 2,4-DNP reagent. This confirms the presence of the carbonyl group. The compound (A) gives yellow precipitate on heating with iodine in the presence of sodium hydroxide. This confirms the presence of methyl keto groups.
The compound (A) neither reduces Tollens' or Fehling's reagent. It confirms the absence of the aldehyde group.
The compound (A) neither decolourises bromine water nor Baeyer's reagent. It confirms the absence of carbon-carbon double or triple bonds.
From this information, you can conclude that the compound is acetophenone.
When you subject acetophenone to vigorous oxidation, you obtain benzoic acid (compound (B) with molecular formula \[{{\text{C}}_{\text{7}}}{{\text{H}}_{\text{6}}}{{\text{O}}_2}\] .

Hence, the compound (A) is acetophenone and the compound (B) is benzoic acid.
Note:
An aldehyde (or a ketone) reacts with 2,4-DNP to form a 2,4-dinitrophenyl hydrazone derivative. Here, 2,4-DNP reagent means 2,4-dinitrophenyl hydrazine. Aldehydes are oxidized by Tollen’s reagent to give black silver mirror. Tollen’s reagent is an ammoniacal silver nitrate solution. Ketones do not react with Tollen’s reagent. Silver mirror test is used to distinguish between aldehydes and ketones.
Recently Updated Pages
Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Master Class 12 Business Studies: Engaging Questions & Answers for Success

Trending doubts
How many chromosomes are found in human ovum a 46 b class 12 biology CBSE

The diagram of the section of a maize grain is given class 12 biology CBSE

No of 5 membered ring in left Caleft EDTA right right2 class 12 chemistry CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE

How many 176Omega resistors in parallel are required class 12 physics CBSE

What is corrosion Explain its advantages and disad class 12 chemistry CBSE




