
An organic compound (A) with molecular formula ${C_7}{H_8}O$ dissolves in $NaOH$ and gives characteristic color with $FeC{l_3}$ . On treatment with $B{r_2}$ , it gives a tribromo product ${C_7}{H_5}OB{r_3}$ . The compound is –
A. p-hydroxybenzene
B. 2-methoxy-2-phenyl propane
C. m-cresol
D. p-cresol
Answer
573.9k+ views
Hint:T First try to find the type of compound from the given formula. $FeC{l_3}$ is used to detect the presence of phenol in the compound. The number of sites of unsaturation can also be found out. Reaction of bromine to give a trisubstituted product is an example of electrophilic substitution reaction.
Complete step by step answer:
The structures of the given compounds are-
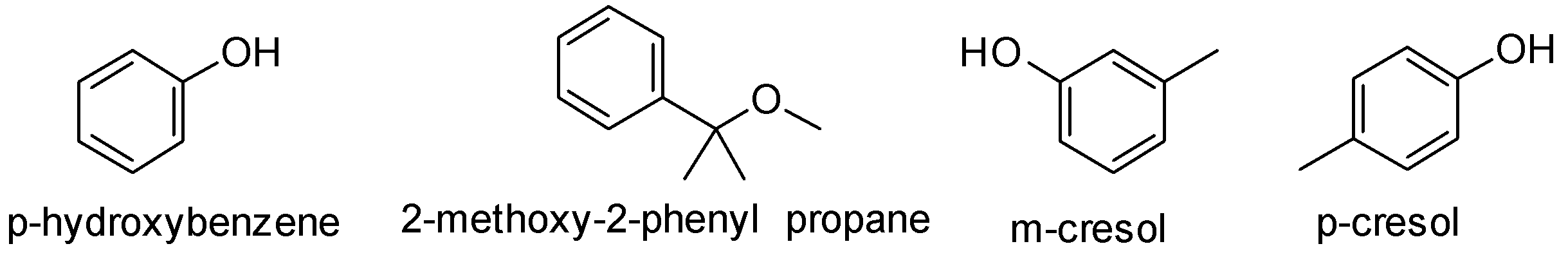
The formula given suggests that there are four sites of unsaturation in the given compound A. It can be calculated as-
Formula- ${C_7}{H_8}O$
According to the $2n + 2$ rule,
$n = 7$ , the saturated compound will have formula ${C_7}{H_{16}}$ .Here we do not count anything for oxygen atoms. So there are eight hydrogen atoms less. It means that there are four sites of unsaturation in the given sample compound. We could suggest the presence of an aromatic ring that has four sites of unsaturation.
Compound (A) gives color with $FeC{l_3}$ . It is used to detect the presence of phenols in the sample. So this confirms that our given compound (A) is a phenol. 2-methoxy-2-phenylpropane is an ether. Also it has eleven carbon atoms while compound (A) has only six carbon atoms. So option B cannot be the right answer and can be ruled out.
Also p-hydroxybenzene has only six carbon atoms while the given compound (A) has seven carbon atoms. So option A is not the correct answer.
Compound (A) on reaction with bromine gives a tribromo product. Phenols undergo electrophilic substitution reactions. The $ - OH$ group activates the benzene ring and makes the ortho, para position electron rich due to resonance effect. Hence the incoming group is directed to ortho and para position. The tribromo product suggests that the three bromine atoms must be present at the ortho and para position. m-cresol has methyl group at meta position and the ortho and para positions are vacant. So it can form a tribromo product.
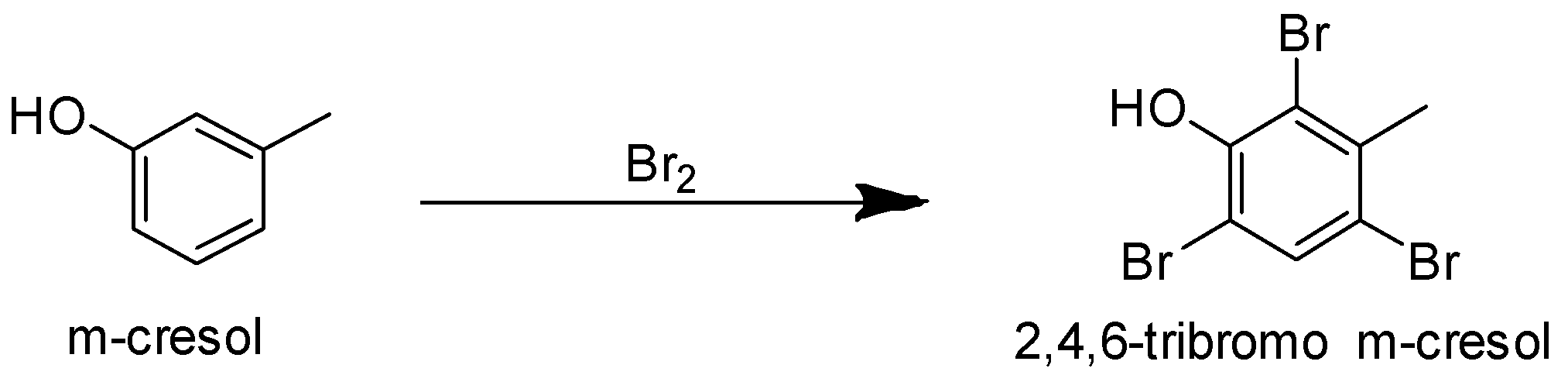
p-cresol cannot form a tribromo product with bromine as it has methyl group at its para position. So the para position is blocked for the incoming bromine atom. Bromine can be substituted only at the ortho position.
Hence correct answer is optionC.
Note:
Phenols undergo electrophilic substitution reactions at the ortho and para positions since the $ - OH$ group is an activating group.
$FeC{l_3}$ is used as a test for detection of phenols. If phenols are present in the sample they give a characteristic color.
Halogens and $ - OH$ groups are electron releasing groups. They give ortho-para substituted products.
Complete step by step answer:
The structures of the given compounds are-

The formula given suggests that there are four sites of unsaturation in the given compound A. It can be calculated as-
Formula- ${C_7}{H_8}O$
According to the $2n + 2$ rule,
$n = 7$ , the saturated compound will have formula ${C_7}{H_{16}}$ .Here we do not count anything for oxygen atoms. So there are eight hydrogen atoms less. It means that there are four sites of unsaturation in the given sample compound. We could suggest the presence of an aromatic ring that has four sites of unsaturation.
Compound (A) gives color with $FeC{l_3}$ . It is used to detect the presence of phenols in the sample. So this confirms that our given compound (A) is a phenol. 2-methoxy-2-phenylpropane is an ether. Also it has eleven carbon atoms while compound (A) has only six carbon atoms. So option B cannot be the right answer and can be ruled out.
Also p-hydroxybenzene has only six carbon atoms while the given compound (A) has seven carbon atoms. So option A is not the correct answer.
Compound (A) on reaction with bromine gives a tribromo product. Phenols undergo electrophilic substitution reactions. The $ - OH$ group activates the benzene ring and makes the ortho, para position electron rich due to resonance effect. Hence the incoming group is directed to ortho and para position. The tribromo product suggests that the three bromine atoms must be present at the ortho and para position. m-cresol has methyl group at meta position and the ortho and para positions are vacant. So it can form a tribromo product.

p-cresol cannot form a tribromo product with bromine as it has methyl group at its para position. So the para position is blocked for the incoming bromine atom. Bromine can be substituted only at the ortho position.
Hence correct answer is optionC.
Note:
Phenols undergo electrophilic substitution reactions at the ortho and para positions since the $ - OH$ group is an activating group.
$FeC{l_3}$ is used as a test for detection of phenols. If phenols are present in the sample they give a characteristic color.
Halogens and $ - OH$ groups are electron releasing groups. They give ortho-para substituted products.
Recently Updated Pages
Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 12 English: Engaging Questions & Answers for Success

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Business Studies: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

What are the major means of transport Explain each class 12 social science CBSE

Draw a labelled sketch of the human eye class 12 physics CBSE

Why cannot DNA pass through cell membranes class 12 biology CBSE

Differentiate between insitu conservation and exsitu class 12 biology CBSE

Draw a neat and well labeled diagram of TS of ovary class 12 biology CBSE




