An organic Compound (A) is widely used as a preservative in a pickle and has a molecular formula \[{{\text{C}}_{\text{2}}}{{\text{H}}_{\text{4}}}{{\text{O}}_{\text{2}}}\]. This compound reacts with ethanol to form a sweet smelling compound (B)
A.Identify the compounds A and B
B.Name the process and write the corresponding chemical equation
Answer
612k+ views
Hint:It is a carboxylic acid to which methyl group is attached.The other name of compound (A) formed is Vinegar, and vinegar is used as preservative in pickle. Molecular Weight of acetic acid is \[{\text{60}}{\text{.052g/mol}}\].
Complete step by step answer:
We know Ethanoic acid is used as a solvent and as a Vinegar in the food Industry, vinegar is the dilute solution of acetic acid. Its structural formula is \[{\text{CH3COOH}}\], therefore compound (A) is Acetic acid.
Also when compound (A) reacts with Ethanol it produces sweet smelling compound which is an ester i.e.,
\[{\text{CH3COOH}}\]\[ + {{\text{C}}_{\text{2}}}{{\text{H}}_{\text{5}}}{\text{OH}}\mathop \leftrightarrows \limits^{{H^ + }} {\text{C}}{{\text{H}}_{\text{3}}}{\text{COO}}{{\text{C}}_{\text{2}}}{{\text{H}}_{\text{5}}} + {{\text{H}}_2}{\text{O}}\]
Hence the compound (B) formed is Ethyl Ethanoate.
To get ethanoic acid from ethyl ethanoate hydrolysis process is followed with the chemical equation
${\text{C}}{{\text{H}}_{\text{3}}}{\text{COO}}{{\text{C}}_{\text{2}}}{{\text{H}}_{\text{5}}}{\text{ + }}{{\text{H}}_{\text{2}}}{\text{O}}\xrightarrow{{{{\text{H}}^{\text{ + }}}}}{\text{C}}{{\text{H}}_3}{\text{COOH + }}{{\text{C}}_2}{{\text{H}}_5}{\text{OH}}$
Additional Information- Ethanoic acid, also called acetic acid as its common name. . It is used in making a polymer called acetate rayon.The structure of ethanoic acid is
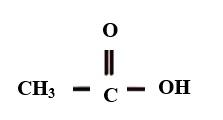
Ethanoic acid is when it gets undiluted it is known as glacial acid.
Note:
Ethanoic acid, also called acetic acid as its common name, is a weak acid distinguishable by its sharp, vinegar like smell .
In Chemistry, after every reaction has thus formed will always give a foul smell, Only Ester is a compound in general which gives a fruity smell/pleasant smell. It is also used in the food industry as an acidity regulator.
Complete step by step answer:
We know Ethanoic acid is used as a solvent and as a Vinegar in the food Industry, vinegar is the dilute solution of acetic acid. Its structural formula is \[{\text{CH3COOH}}\], therefore compound (A) is Acetic acid.
Also when compound (A) reacts with Ethanol it produces sweet smelling compound which is an ester i.e.,
\[{\text{CH3COOH}}\]\[ + {{\text{C}}_{\text{2}}}{{\text{H}}_{\text{5}}}{\text{OH}}\mathop \leftrightarrows \limits^{{H^ + }} {\text{C}}{{\text{H}}_{\text{3}}}{\text{COO}}{{\text{C}}_{\text{2}}}{{\text{H}}_{\text{5}}} + {{\text{H}}_2}{\text{O}}\]
Hence the compound (B) formed is Ethyl Ethanoate.
To get ethanoic acid from ethyl ethanoate hydrolysis process is followed with the chemical equation
${\text{C}}{{\text{H}}_{\text{3}}}{\text{COO}}{{\text{C}}_{\text{2}}}{{\text{H}}_{\text{5}}}{\text{ + }}{{\text{H}}_{\text{2}}}{\text{O}}\xrightarrow{{{{\text{H}}^{\text{ + }}}}}{\text{C}}{{\text{H}}_3}{\text{COOH + }}{{\text{C}}_2}{{\text{H}}_5}{\text{OH}}$
Additional Information- Ethanoic acid, also called acetic acid as its common name. . It is used in making a polymer called acetate rayon.The structure of ethanoic acid is
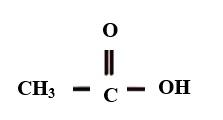
| Molecular Weight: \[{\text{62}}{\text{.037 }}\dfrac{{\text{g}}}{{{\text{mol}}}}\] |
Ethanoic acid is when it gets undiluted it is known as glacial acid.
Note:
Ethanoic acid, also called acetic acid as its common name, is a weak acid distinguishable by its sharp, vinegar like smell .
In Chemistry, after every reaction has thus formed will always give a foul smell, Only Ester is a compound in general which gives a fruity smell/pleasant smell. It is also used in the food industry as an acidity regulator.
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Trending doubts
Which is more stable and why class 12 chemistry CBSE

Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

What are the major means of transport Explain each class 12 social science CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE




