An example of a secondary alcohol is:
A)1-propanol
B)2-propanol
C)1,2-propanol
D)1,2,3-propanol
Answer
615.9k+ views
Hint: To answer this answer, first we need to know what secondary alcohol is. A secondary alcohol is where the alcohol is placed which is bonded to two other carbon atoms.
Complete step-by-step answer:
-In chemistry, alcohol groups are those that in a compound there is at least one hydroxyl group attached. This hydroxyl group is the functional group of the compound.
-The alcohol groups are divided into three types on the basis of the total number of alkyl groups connected to the carbon connected to the hydroxyl group.
-Now, let us discuss the nomenclature of these compounds according to IUPAC.
-The primary alcohols have general formulas $\mathrm{RCH}_{2} \mathrm{OH}$. The simplest primary alcohol is methanol $\left(\mathrm{CH}_{3} \mathrm{OH}\right),$ for which $\mathrm{R}=\mathrm{H},$ and the next is ethanol, for which $\mathrm{R}=\mathrm{CH}_{3},$ the methyl group. Secondary alcohols are those of the form $\mathrm{RR}^{\prime} \mathrm{CHOH},$ the simplest of which is 2 -propanol $\left(\mathrm{R}=\mathrm{R}^{\prime}=\mathrm{CH}_{3}\right) .$ For the tertiary alcohols the general form is $\mathrm{RR}^{\prime} \mathrm{R}^{\prime \prime} \mathrm{COH} .$ The simplest example is tert-butanol ( 2-methylpropan-2-ol), for which each of R, R', and R" is CH $_{3}$. In these shorthands, $\mathrm{R}, \mathrm{R}^{\prime},$ and $\mathrm{R}^{\prime \prime}$ represent substituents, alkyl or other attached, generally organic groups.
-Discussing option A, we have 1 propanol. So, according to the IUPAC the number of carbon will start from the right one. But, as we can see that there is only one carbon attached to the hydroxyl group. Hence, it is primary alcohol.
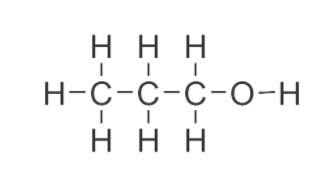
-Discussing option B, we have 2 propanol.The hydroxyl group is attached to a secondary C atom (C atom bearing only one H atom). Thus, it is clear that it is a secondary alcohol.
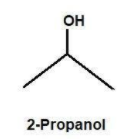
Clearly the answer is B.
Note: Always look out for the secondary carbon atom in the compound which bears the hydroxyl group. That is how, it is a secondary alcohol. A chemical test called Lucas test is also done to distinguish between the primary,secondary and tertiary alcohol. Primary alcohol does not respond to the test. Secondary takes a bit of time and gives the result, while the tertiary alcohols are the fastest in this test.
Complete step-by-step answer:
-In chemistry, alcohol groups are those that in a compound there is at least one hydroxyl group attached. This hydroxyl group is the functional group of the compound.
-The alcohol groups are divided into three types on the basis of the total number of alkyl groups connected to the carbon connected to the hydroxyl group.
-Now, let us discuss the nomenclature of these compounds according to IUPAC.
-The primary alcohols have general formulas $\mathrm{RCH}_{2} \mathrm{OH}$. The simplest primary alcohol is methanol $\left(\mathrm{CH}_{3} \mathrm{OH}\right),$ for which $\mathrm{R}=\mathrm{H},$ and the next is ethanol, for which $\mathrm{R}=\mathrm{CH}_{3},$ the methyl group. Secondary alcohols are those of the form $\mathrm{RR}^{\prime} \mathrm{CHOH},$ the simplest of which is 2 -propanol $\left(\mathrm{R}=\mathrm{R}^{\prime}=\mathrm{CH}_{3}\right) .$ For the tertiary alcohols the general form is $\mathrm{RR}^{\prime} \mathrm{R}^{\prime \prime} \mathrm{COH} .$ The simplest example is tert-butanol ( 2-methylpropan-2-ol), for which each of R, R', and R" is CH $_{3}$. In these shorthands, $\mathrm{R}, \mathrm{R}^{\prime},$ and $\mathrm{R}^{\prime \prime}$ represent substituents, alkyl or other attached, generally organic groups.
-Discussing option A, we have 1 propanol. So, according to the IUPAC the number of carbon will start from the right one. But, as we can see that there is only one carbon attached to the hydroxyl group. Hence, it is primary alcohol.

-Discussing option B, we have 2 propanol.The hydroxyl group is attached to a secondary C atom (C atom bearing only one H atom). Thus, it is clear that it is a secondary alcohol.

Clearly the answer is B.
Note: Always look out for the secondary carbon atom in the compound which bears the hydroxyl group. That is how, it is a secondary alcohol. A chemical test called Lucas test is also done to distinguish between the primary,secondary and tertiary alcohol. Primary alcohol does not respond to the test. Secondary takes a bit of time and gives the result, while the tertiary alcohols are the fastest in this test.
Recently Updated Pages
Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 English: Engaging Questions & Answers for Success

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 12 Business Studies: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

Why is the cell called the structural and functional class 12 biology CBSE

Draw ray diagrams each showing i myopic eye and ii class 12 physics CBSE

Which is the correct genotypic ratio of mendel dihybrid class 12 biology CBSE




