An equimolar-quantities of ethanol and propanol is heated with conc. ${{H}_{2}}S{{O}_{4}}$. The product formed is/are?
(A) ${{C}_{2}}{{H}_{5}}O{{C}_{2}}{{H}_{5}}$
(B) ${{C}_{3}}{{H}_{7}}O{{C}_{3}}{{H}_{7}}$
(C) ${{C}_{2}}{{H}_{5}}O{{C}_{3}}{{H}_{7}}$
(D) All of these
Answer
602.4k+ views
Hint: As two types of alcohol are involved in the reaction, formation of only a single product is impossible.
Complete step by step solution:
As, we can see that the equations are all ethers. Thus, formation of ether is involved when alcohol is heated with conc. ${{H}_{2}}S{{O}_{4}}$.
Etherification-
Alcohols undergo dehydration to produce alkenes and ethers (at some special conditions) in the presence of ${{H}_{2}}S{{O}_{4}}$.Formation of ether from the dehydration of alcohol is a nucleophilic substitution reaction. Generally,
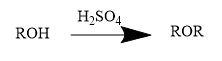
Thus,
When alcohol is heated with conc. Sulphuric acid, the reaction follows two steps of mechanism.
1. Formation of alkyl hydrogen sulphate.
2. Formation of dialkyl ether.
The large amount of alcohol can be used in the conversion of ether with small quantities of conc. ${{H}_{2}}S{{O}_{4}}$ as it can be reused in the following processes. Hence, this is called the continuous etherification reaction.
Illustration-
When an equimolar-quantities of ethanol and propanol are heated with conc. ${{H}_{2}}S{{O}_{4}}$ then,
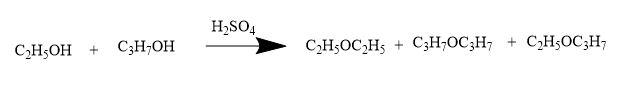
Thus, the products formed are,
${{C}_{2}}{{H}_{5}}O{{C}_{2}}{{H}_{5}}$, ${{C}_{3}}{{H}_{7}}O{{C}_{3}}{{H}_{7}}$, ${{C}_{2}}{{H}_{5}}O{{C}_{3}}{{H}_{7}}$.
Therefore, option (D) is correct.
Note: When the groups of alcohol are different but are reacting with each other in the presence of conc. ${{H}_{2}}S{{O}_{4}}$ then the ether formed will be of three types including groups separately and in combination as well. Ether are ester and would be visible the same when we see the chemical formula. But do remember that they differ as esters have double bonded oxygen within the C atoms and ethers have a single bond. Do not get confused just by looking at the options.
Complete step by step solution:
As, we can see that the equations are all ethers. Thus, formation of ether is involved when alcohol is heated with conc. ${{H}_{2}}S{{O}_{4}}$.
Etherification-
Alcohols undergo dehydration to produce alkenes and ethers (at some special conditions) in the presence of ${{H}_{2}}S{{O}_{4}}$.Formation of ether from the dehydration of alcohol is a nucleophilic substitution reaction. Generally,

Thus,
When alcohol is heated with conc. Sulphuric acid, the reaction follows two steps of mechanism.
1. Formation of alkyl hydrogen sulphate.
2. Formation of dialkyl ether.
The large amount of alcohol can be used in the conversion of ether with small quantities of conc. ${{H}_{2}}S{{O}_{4}}$ as it can be reused in the following processes. Hence, this is called the continuous etherification reaction.
Illustration-
When an equimolar-quantities of ethanol and propanol are heated with conc. ${{H}_{2}}S{{O}_{4}}$ then,

Thus, the products formed are,
${{C}_{2}}{{H}_{5}}O{{C}_{2}}{{H}_{5}}$, ${{C}_{3}}{{H}_{7}}O{{C}_{3}}{{H}_{7}}$, ${{C}_{2}}{{H}_{5}}O{{C}_{3}}{{H}_{7}}$.
Therefore, option (D) is correct.
Note: When the groups of alcohol are different but are reacting with each other in the presence of conc. ${{H}_{2}}S{{O}_{4}}$ then the ether formed will be of three types including groups separately and in combination as well. Ether are ester and would be visible the same when we see the chemical formula. But do remember that they differ as esters have double bonded oxygen within the C atoms and ethers have a single bond. Do not get confused just by looking at the options.
Recently Updated Pages
Three beakers labelled as A B and C each containing 25 mL of water were taken A small amount of NaOH anhydrous CuSO4 and NaCl were added to the beakers A B and C respectively It was observed that there was an increase in the temperature of the solutions contained in beakers A and B whereas in case of beaker C the temperature of the solution falls Which one of the following statements isarecorrect i In beakers A and B exothermic process has occurred ii In beakers A and B endothermic process has occurred iii In beaker C exothermic process has occurred iv In beaker C endothermic process has occurred

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

What are the major means of transport Explain each class 12 social science CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE

Why should a magnesium ribbon be cleaned before burning class 12 chemistry CBSE




