An aqueous solution of A gives yellow precipitate with $K_2Cr_2O_4$. A may contain
A.$P{b^{2 + }}$ or $A{g^ + }$ or $B{a^{2 + }}$
B.$P{b^{2 + }}$or $B{a^{2 + }}$
C.$A{g^ + }$ or $B{a^{2 + }}$
D.$P{b^{2 + }}$ or $A{g^ + }$
Answer
591.3k+ views
Hint: The inorganic ions are categorized into different groups for their qualitative analysis. Inorganic salts consist of two parts known as radicals. The positively charged part (cation) is called a basic radical and the negatively charged part of the salt (anion) is called an acidic radical.
Complete step by step answer:
-Qualitative analysis involves the identification of cations and anions which are present in the salt or a mixture of salts. In this type of analysis we try to find out the nature of substance and the identity of the constituents.
-Inorganic qualitative analysis mainly involves the identification of cations and anions with the help of various reagents. The cations are mainly categorized into six different groups. There is systematic analysis.
-Potassium chromate $left( {{K_2}Cr{O_4}} right)$ is used to detect the presence of $P{b^{2 + }}$ and [B{a^{2 + }}]ions.
-In the first group lead is precipitated as lead chloride. On adding a potassium chromate solution, a yellow precipitate of lead chromate is obtained. This confirms the presence of lead ions.
$PbC{l_2}\, + \,{K_2}Cr{O_4} \to PbCr{O_4}\, + \,2KCl$
-The group five cations are precipitated as their carbonates which dissolve in acetic acid due to the formation of corresponding acetates.
-Potassium chromate solution gives a yellow precipitate of barium carbonate when the solution of fifth group precipitate in acetic acid is treated with it.
$BaC{O_3}\, + \,C{H_3}COOH\, \to {(C{H_3}COOH)_2}Ba\, + \,{H_2}O\, + \,C{O_2}$
${(C{H_3}COO)_2}Ba\, + \,{K_2}Cr{O_4}\, \to BaCr{O_4}\, + \,2C{H_3}COOK$
-So we can see that lead and barium ions are detected with the help of potassium chromate solution.
Hence the correct option is B.
Note:
-The structure of potassium chromate is
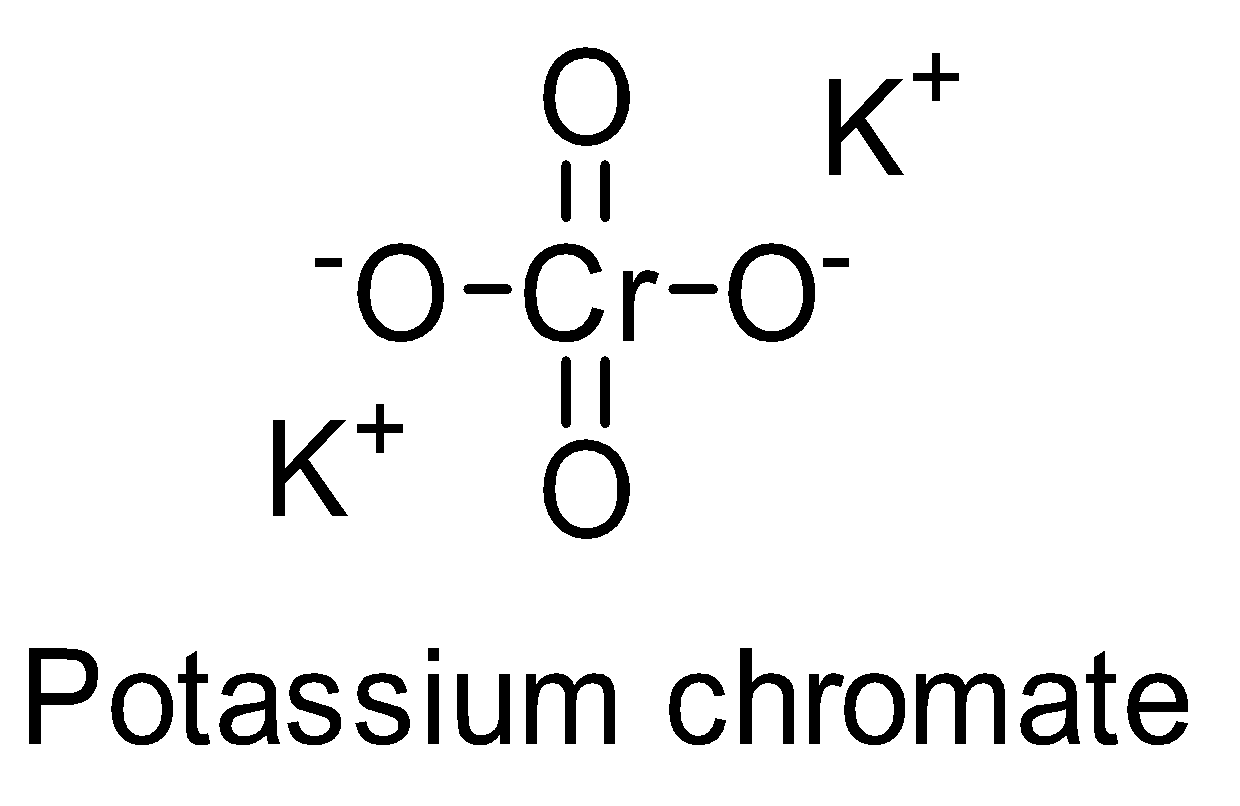
-Potassium chromate is a water-soluble compound. It appears as a yellow crystalline solid. Its mineral form is called tarapacaite.
Complete step by step answer:
-Qualitative analysis involves the identification of cations and anions which are present in the salt or a mixture of salts. In this type of analysis we try to find out the nature of substance and the identity of the constituents.
-Inorganic qualitative analysis mainly involves the identification of cations and anions with the help of various reagents. The cations are mainly categorized into six different groups. There is systematic analysis.
-Potassium chromate $left( {{K_2}Cr{O_4}} right)$ is used to detect the presence of $P{b^{2 + }}$ and [B{a^{2 + }}]ions.
-In the first group lead is precipitated as lead chloride. On adding a potassium chromate solution, a yellow precipitate of lead chromate is obtained. This confirms the presence of lead ions.
$PbC{l_2}\, + \,{K_2}Cr{O_4} \to PbCr{O_4}\, + \,2KCl$
-The group five cations are precipitated as their carbonates which dissolve in acetic acid due to the formation of corresponding acetates.
-Potassium chromate solution gives a yellow precipitate of barium carbonate when the solution of fifth group precipitate in acetic acid is treated with it.
$BaC{O_3}\, + \,C{H_3}COOH\, \to {(C{H_3}COOH)_2}Ba\, + \,{H_2}O\, + \,C{O_2}$
${(C{H_3}COO)_2}Ba\, + \,{K_2}Cr{O_4}\, \to BaCr{O_4}\, + \,2C{H_3}COOK$
-So we can see that lead and barium ions are detected with the help of potassium chromate solution.
Hence the correct option is B.
Note:
-The structure of potassium chromate is

-Potassium chromate is a water-soluble compound. It appears as a yellow crystalline solid. Its mineral form is called tarapacaite.
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Trending doubts
Which is more stable and why class 12 chemistry CBSE

Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

What are the major means of transport Explain each class 12 social science CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE




