An alcoholic solution of dimethylglyoxime is added to an aqueous solution of nickel (II) chloride. Slow addition of ammonium hydroxide leads to the precipitation of a rosy red coloured metal complex. Then find out the number of hydrogen bonds present in the structure of the complex.
(A) 2
(B) 3
(C) 3
(D) None
Answer
568.5k+ views
Hint: The red coloured complex is the product in the form of precipitate which is produced when an alcoholic solution of dimethylglyoxime is added to an aqueous solution of nickel (II) chloride.
Also, it’s a hint that the valency of nickel will determine the complex formation and the number of hydrogen bonds formed.
Complete step-by-step answer: Let us discuss the reactants involved to form a product whose hydrogen bonds needs to be counted;
Dimethylglyoxime is the whitish crystalline solid with molecular formula of ${{C}_{4}}{{H}_{8}}{{N}_{2}}{{O}_{2}}$ (also known as 2, 3 – butadiene dioxide).
When $dmg{{H}_{2}}$ reacts with metals salts, a complex is formed i.e. metal-dmg is formed. So, in the case of nickel (II) chloride; the following displayed complex is formed.
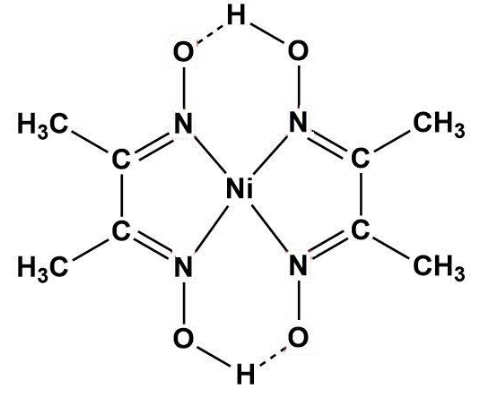
Here, we can see that when $N{{i}^{2+}}$ reacts with $dmg{{H}_{2}}$, a Ni-dmg complex is formed having two hydrogen bonds (also as the valency of nickel is 2 in ionic form).
Therefore option (2) is correct.
Note:Dimethylglyoxime is actually used for the analysis and identification of nickel and palladium.
Specifically, for the nickel atom, the white coloured powder changes to a rosy red colour complex; hence, the presence of nickel is determined.
Also, it’s a hint that the valency of nickel will determine the complex formation and the number of hydrogen bonds formed.
Complete step-by-step answer: Let us discuss the reactants involved to form a product whose hydrogen bonds needs to be counted;
Dimethylglyoxime is the whitish crystalline solid with molecular formula of ${{C}_{4}}{{H}_{8}}{{N}_{2}}{{O}_{2}}$ (also known as 2, 3 – butadiene dioxide).
When $dmg{{H}_{2}}$ reacts with metals salts, a complex is formed i.e. metal-dmg is formed. So, in the case of nickel (II) chloride; the following displayed complex is formed.

Here, we can see that when $N{{i}^{2+}}$ reacts with $dmg{{H}_{2}}$, a Ni-dmg complex is formed having two hydrogen bonds (also as the valency of nickel is 2 in ionic form).
Therefore option (2) is correct.
Note:Dimethylglyoxime is actually used for the analysis and identification of nickel and palladium.
Specifically, for the nickel atom, the white coloured powder changes to a rosy red colour complex; hence, the presence of nickel is determined.
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Trending doubts
Ribosomal RNA is actively synthesised in A Nucleoplasm class 12 biology CBSE

In a transcription unit the promoter is said to be class 12 biology CBSE

Verhulst Pearl logistic growth described by the equation class 12 biology CBSE

Give reasons for the following Aquatic animals are class 12 chemistry CBSE

A foreign DNA is inserted and lighted at BamHI site class 12 biology CBSE

A human protein that is obtained from transgenic animals class 12 biology CBSE




