
Among the following, which is antiaromatic
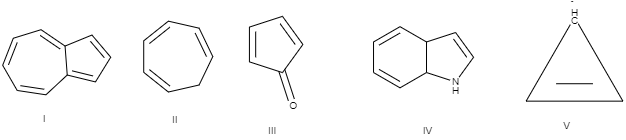
A.\[I\text{ }and\text{ }IV\]
B.\[III\text{ }and\text{ }IV\]
C.\[II\text{ }and\text{ }V\]
D.\[I\text{ }and\text{ }III\]

Answer
590.4k+ views
Hint: Antiaromatic molecules or ions have a characteristic of a cyclic molecule with a pi electron system that are of higher energy due to the presence of delocalised electrons in it.
Unlike the aromatic compounds, which are highly stable and follow Hückel's rule, antiaromatic compounds are highly reactive and unstable and do not follow Huckel’s rule .
Complete step by step answer:
Aromatic compounds are a type of chemical compounds which consist of conjugated planar ring systems along with delocalized pi-electron clouds in place of individual, having alternating double and single bonds.
They are also known as aromatics or arenes. The most common examples are benzene and toluene. For a compound to be Aromatics, it requires satisfying the Huckel’s rule. Microorganisms and Plants have an exclusive route to aromatic compounds having benzene rings. Therefore a huge majority of aromatic compounds in nature are produced by microorganisms and plants, and animals are dependent upon plants for many aromatic compounds either directly or indirectly.
On the other hand, anti-aromatic compounds can be defined as a cyclic compound which doesn’t necessarily have a continuous form of overlapping ring of p orbitals must not be considered as aromatic or even anti aromatic and hence these are known as nonaromatic or aliphatic compounds.
Non-aromatic compounds' electronic energy is like that of its open-chain counterpart. Crude oil comprises mainly alkanes and large releases into the sea due to the wreckage of oil tankers results in the death of many seabirds and other marine organisms because of their physical effects of oiling or smothering with these compounds.
Anti-aromatic compounds are those which are cyclic molecules and have a planar structure in which all the bonded atoms lie in the same plane.
The basic difference between aromatic and antiaromatic compounds is that aromatic compounds include the benzene ring type structures having alternate double bonds but anti aromatic compounds doesn’t.
If we look at the options, B satisfies all the criteria necessary for a compound to be antiaromatic.
Hence the correct answer is option B.
Note:
Non aromatic compounds do not show any resonance in the compound.
Some examples of non aromatic compounds are alkanes, alkenes, alkynes. And that of anti aromatic compounds are cyclobutadiene, cyclohexane etc.
Unlike the aromatic compounds, which are highly stable and follow Hückel's rule, antiaromatic compounds are highly reactive and unstable and do not follow Huckel’s rule .
Complete step by step answer:
Aromatic compounds are a type of chemical compounds which consist of conjugated planar ring systems along with delocalized pi-electron clouds in place of individual, having alternating double and single bonds.
They are also known as aromatics or arenes. The most common examples are benzene and toluene. For a compound to be Aromatics, it requires satisfying the Huckel’s rule. Microorganisms and Plants have an exclusive route to aromatic compounds having benzene rings. Therefore a huge majority of aromatic compounds in nature are produced by microorganisms and plants, and animals are dependent upon plants for many aromatic compounds either directly or indirectly.
On the other hand, anti-aromatic compounds can be defined as a cyclic compound which doesn’t necessarily have a continuous form of overlapping ring of p orbitals must not be considered as aromatic or even anti aromatic and hence these are known as nonaromatic or aliphatic compounds.
Non-aromatic compounds' electronic energy is like that of its open-chain counterpart. Crude oil comprises mainly alkanes and large releases into the sea due to the wreckage of oil tankers results in the death of many seabirds and other marine organisms because of their physical effects of oiling or smothering with these compounds.
Anti-aromatic compounds are those which are cyclic molecules and have a planar structure in which all the bonded atoms lie in the same plane.
The basic difference between aromatic and antiaromatic compounds is that aromatic compounds include the benzene ring type structures having alternate double bonds but anti aromatic compounds doesn’t.
If we look at the options, B satisfies all the criteria necessary for a compound to be antiaromatic.
Hence the correct answer is option B.
Note:
Non aromatic compounds do not show any resonance in the compound.
Some examples of non aromatic compounds are alkanes, alkenes, alkynes. And that of anti aromatic compounds are cyclobutadiene, cyclohexane etc.
Recently Updated Pages
Basicity of sulphurous acid and sulphuric acid are

Master Class 12 English: Engaging Questions & Answers for Success

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Which country did Danny Casey play for class 12 english CBSE

Give 10 examples of unisexual and bisexual flowers

Coming together federation is practiced in A India class 12 social science CBSE

Write the formula to find the shortest distance between class 12 maths CBSE




