Alicyclic compounds are:
a.) Aromatic compounds
b.) Aliphatic compounds
c.) Heterocyclic compounds
d.) Aliphatic cyclic compounds
Answer
632.7k+ views
Hint: Non aromatic cyclic compounds are called alicyclic compounds. Non aromatic compounds are called aliphatic. It may be unicyclic or multicyclic.
Complete step by step solution:
The compounds which have one or more all-carbon rings, either saturated or unsaturated but do not have aromatic character are called alicyclic compounds. In other words, alicyclic compounds are aliphatic cyclic compounds. The bonds between pairs of adjacent atoms may all be of the type designated single bonds (involving two electrons), or some of them may be double or triple bonds (with four or six electrons, respectively); six-membered rings for which a system of alternating single and double bonds may be envisioned. Alicyclic compounds may have one or more aliphatic side chains attached.
The simplest alicyclic compounds are the monocyclic cycloalkanes. Monocyclic cycloalkanes include: cyclopropane, cyclobutene, cyclopentane, cyclohexane, cycloheptane, cyclooctane, etc.
Bicyclic alkanes include: bicyclo undecane, decalin, etc.
Polycyclic alkanes include cubane, basketane and tetrahedrane.
The smallest alicyclic compound is cyclopropane. The mode of ring-closing in the formation of many alicyclic compounds can be predicted by Baldwin's rules.
Those alicyclic compounds in which the ring contains three or four carbon atoms are less stable than the compounds having larger rings, because the angles formed by adjacent covalent bonds are smaller than is necessary for maximum effectiveness. In the larger rings all the bond angles have the preferred value (about 109.5°); consequently, the atoms in the ring do not lie in one plane.
Some structures of alicyclics are:
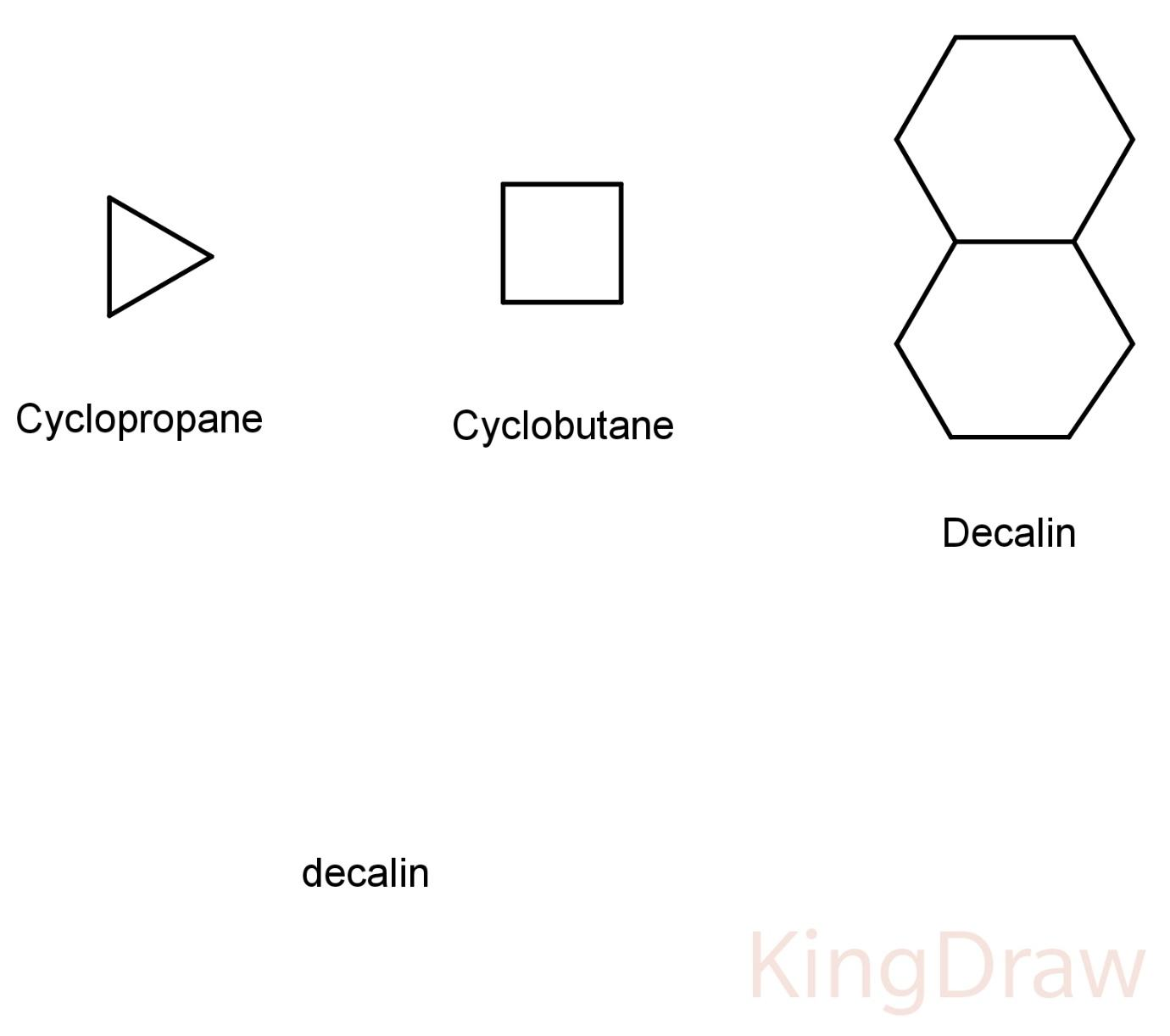
Hence, the correct answer is (D) aliphatic cyclic compounds.
Note: A student must know that homocyclic organic compounds are classified into aromatic and alicyclic compounds. Remember that the alicyclic compounds may have one or more aliphatic side chains attached.
Complete step by step solution:
The compounds which have one or more all-carbon rings, either saturated or unsaturated but do not have aromatic character are called alicyclic compounds. In other words, alicyclic compounds are aliphatic cyclic compounds. The bonds between pairs of adjacent atoms may all be of the type designated single bonds (involving two electrons), or some of them may be double or triple bonds (with four or six electrons, respectively); six-membered rings for which a system of alternating single and double bonds may be envisioned. Alicyclic compounds may have one or more aliphatic side chains attached.
The simplest alicyclic compounds are the monocyclic cycloalkanes. Monocyclic cycloalkanes include: cyclopropane, cyclobutene, cyclopentane, cyclohexane, cycloheptane, cyclooctane, etc.
Bicyclic alkanes include: bicyclo undecane, decalin, etc.
Polycyclic alkanes include cubane, basketane and tetrahedrane.
The smallest alicyclic compound is cyclopropane. The mode of ring-closing in the formation of many alicyclic compounds can be predicted by Baldwin's rules.
Those alicyclic compounds in which the ring contains three or four carbon atoms are less stable than the compounds having larger rings, because the angles formed by adjacent covalent bonds are smaller than is necessary for maximum effectiveness. In the larger rings all the bond angles have the preferred value (about 109.5°); consequently, the atoms in the ring do not lie in one plane.
Some structures of alicyclics are:

Hence, the correct answer is (D) aliphatic cyclic compounds.
Note: A student must know that homocyclic organic compounds are classified into aromatic and alicyclic compounds. Remember that the alicyclic compounds may have one or more aliphatic side chains attached.
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Social Science: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

Why is the cell called the structural and functional class 12 biology CBSE

Draw ray diagrams each showing i myopic eye and ii class 12 physics CBSE

Which is the correct genotypic ratio of mendel dihybrid class 12 biology CBSE




