Alanine forms Zwitter ion which exists as:
(a)- $C{{H}_{3}}-\overset{\oplus N{{H}_{3}}}{\mathop{\overset{|}{\mathop{C}}\,}}\,-HCO{{O}^{-}}$ in acidic medium
(b)- $C{{H}_{3}}-\overset{\oplus N{{H}_{3}}}{\mathop{\overset{|}{\mathop{C}}\,}}\,-HCOOH$ in a medium of pH = 4
(c)- $C{{H}_{3}}-\underset{N{{H}_{2}}}{\mathop{\underset{|}{\mathop{C}}\,}}\,-HCO{{O}^{-}}$ in a medium of pH = 1
(d)- $C{{H}_{3}}-\underset{N{{H}_{2}}}{\mathop{\underset{|}{\mathop{C}}\,}}\,-HCO{{O}^{-}}$ in a medium of pH = 2
Answer
571.5k+ views
Hint:Alanine is a compound that belongs to amino acids which means the compound will have one amino group and one carboxylic acid group. Zwitter ion is formed when a proton travels within the molecule.
Complete step-by-step answer:We know that amino acids are those compounds in which the same molecule has one amino group, i.e., $(-N{{H}_{2}})$ and one carboxylic acid group, i.e., $(-COOH)$. On specifying alpha-amino acid means the amino group is on the alpha position of the carboxylic acid and alanine is an example of alpha-amino acid group. The molecular formula of alanine is ${{C}_{3}}{{H}_{7}}N{{O}_{2}}$. It is a compound of two carbon atoms in which the same carbon atom has an amine group as well as a carboxylic acid group. The structure of alanine is given below:
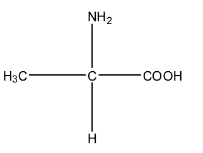
Zwitter ion is formed when a proton travels within the molecule. So, in alanine, the proton from the carboxylic acid group will shift to the amine group because the carboxylic acid has the tendency to release the proton, and amine being base has the tendency to accept the proton. The zwitter ion is formed in an acidic medium. The structure is given below:
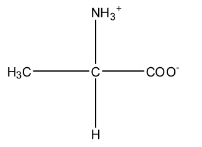
So, from the given options only option (a) resembles the structure of the zwitterion.
Therefore, the correct answer is an option (a).
Note: Zwitterion is only possible if the same molecule has both an electron donor and an electron acceptor. Alanine is a white powder whose molecular formula is 89.094 g/8mol.
Complete step-by-step answer:We know that amino acids are those compounds in which the same molecule has one amino group, i.e., $(-N{{H}_{2}})$ and one carboxylic acid group, i.e., $(-COOH)$. On specifying alpha-amino acid means the amino group is on the alpha position of the carboxylic acid and alanine is an example of alpha-amino acid group. The molecular formula of alanine is ${{C}_{3}}{{H}_{7}}N{{O}_{2}}$. It is a compound of two carbon atoms in which the same carbon atom has an amine group as well as a carboxylic acid group. The structure of alanine is given below:

Zwitter ion is formed when a proton travels within the molecule. So, in alanine, the proton from the carboxylic acid group will shift to the amine group because the carboxylic acid has the tendency to release the proton, and amine being base has the tendency to accept the proton. The zwitter ion is formed in an acidic medium. The structure is given below:

So, from the given options only option (a) resembles the structure of the zwitterion.
Therefore, the correct answer is an option (a).
Note: Zwitterion is only possible if the same molecule has both an electron donor and an electron acceptor. Alanine is a white powder whose molecular formula is 89.094 g/8mol.
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 9 General Knowledge: Engaging Questions & Answers for Success

Master Class 9 Maths: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Name the crygenes that control cotton bollworm and class 12 biology CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

In a transcription unit the promoter is said to be class 12 biology CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE




