According to the MO theory:
A.\[{O_2}^ + \] is paramagnetic and bond order is greater than \[{O_2}\]
B.\[{O_2}^ + \] is paramagnetic and bond order is less than \[{O_2}\]
C.\[{O_2}^ + \] is diamagnetic and bond order is less than \[{O_2}\]
D.\[{O_2}^ + \] is diamagnetic and bond order is more than \[{O_2}\]
Answer
519.9k+ views
Hint: The bond order is a measure of the number of bonds that exist between two atoms and can be calculated by multiplying the difference of number of electrons present in bonding and antibonding molecular orbitals by half.
Complete answer:
A simple oxygen molecule is diatomic in nature and consists of double bonds. The double bond present in oxygen molecules is an indicator of the fact that its bond order is \[2\] .
\[{O_2}^ + \] is chemically different from the simple oxygen molecule \[{O_2}\] as it has lost one electron or it has one electron less as compared to the nitrogen molecule.
The placement of electrons in bonding or antibonding molecular orbitals can be determined by drawing the molecular orbital diagram of the molecule. This diagram is the virtual representation of the linear combination of atomic orbitals that combine to give molecular orbitals.
The molecular orbital diagram of an \[{O_2}^ + \] ion can be shown as follows:
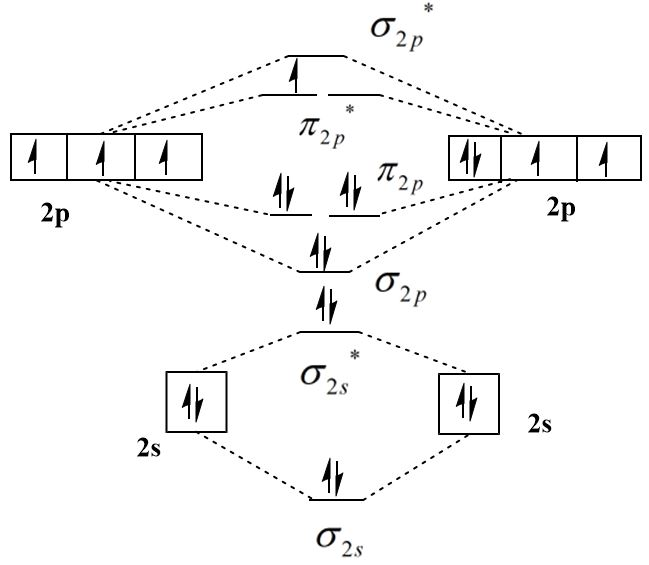
Thus, the outermost electronic configuration of \[{O_2}^ + \] molecule is \[{\sigma _{2s}}^2{\sigma ^*}{_{2s}^2}{\sigma _{2p}}^2{\pi _{2p}}^4{\sigma ^*}{_{2p}^1}\] which indicates that there are a total of eight electrons in bonding molecular orbitals and two electrons in the antibonding molecular orbitals. The following formula can be used to calculate the bond order:
\[{\text{bond order}} = \dfrac{{{\text{bonding electrons}} - anti{\text{ - }}bonding{\text{ electrons}}}}{2}\]
\[{\text{bond order}} = \dfrac{{8 - 3}}{2} = 2.5\]
\[ \Rightarrow \] Thus, the bond order of \[{O_2}^ + \] ion is higher than that of \[{O_2}\] molecule and the presence of an unpaired electron in the highest occupied molecular orbital (HOMO) makes it paramagnetic.
Therefore option (A) is correct.
Note:
The star marked molecular orbitals are the anti-bonding molecular orbitals that are formed due to the destructive overlap that takes place between atomic orbitals. The electrons present in anti-bonding molecular orbital do not contribute in the bond formation and are therefore subtracted.
Complete answer:
A simple oxygen molecule is diatomic in nature and consists of double bonds. The double bond present in oxygen molecules is an indicator of the fact that its bond order is \[2\] .
\[{O_2}^ + \] is chemically different from the simple oxygen molecule \[{O_2}\] as it has lost one electron or it has one electron less as compared to the nitrogen molecule.
The placement of electrons in bonding or antibonding molecular orbitals can be determined by drawing the molecular orbital diagram of the molecule. This diagram is the virtual representation of the linear combination of atomic orbitals that combine to give molecular orbitals.
The molecular orbital diagram of an \[{O_2}^ + \] ion can be shown as follows:

Thus, the outermost electronic configuration of \[{O_2}^ + \] molecule is \[{\sigma _{2s}}^2{\sigma ^*}{_{2s}^2}{\sigma _{2p}}^2{\pi _{2p}}^4{\sigma ^*}{_{2p}^1}\] which indicates that there are a total of eight electrons in bonding molecular orbitals and two electrons in the antibonding molecular orbitals. The following formula can be used to calculate the bond order:
\[{\text{bond order}} = \dfrac{{{\text{bonding electrons}} - anti{\text{ - }}bonding{\text{ electrons}}}}{2}\]
\[{\text{bond order}} = \dfrac{{8 - 3}}{2} = 2.5\]
\[ \Rightarrow \] Thus, the bond order of \[{O_2}^ + \] ion is higher than that of \[{O_2}\] molecule and the presence of an unpaired electron in the highest occupied molecular orbital (HOMO) makes it paramagnetic.
Therefore option (A) is correct.
Note:
The star marked molecular orbitals are the anti-bonding molecular orbitals that are formed due to the destructive overlap that takes place between atomic orbitals. The electrons present in anti-bonding molecular orbital do not contribute in the bond formation and are therefore subtracted.
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE

Why is the cell called the structural and functional class 12 biology CBSE

Which is the correct genotypic ratio of mendel dihybrid class 12 biology CBSE




