According to IUPAC convention, name of the following compound is:
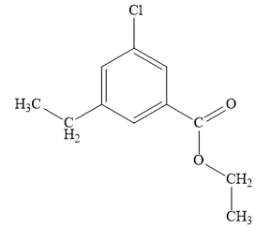
A. 3-chloro-5-ethyl benzoate
B. ethyl-3-chloro-5-ethyl benzoate
C. meta chloro-meta methyl ethyl ethyl benzoate
D. both 'B' and 'C' are correct

Answer
593.7k+ views
Hint:Identify the parent compound and the substituents present. Identify the order of priority of groups for numbering the benzene ring.
Complete answer:
The option A is incorrect because it says that the alkyl part of the alcohol from which benzoate easter was obtained, contains chloro substituent which is incorrect.
The option C is incorrect because in the IUPAC system, the prefix meta is not used. Instead the carbon atoms of benzene ring are numbered. So, you can rule out both options C and D.
Draw the structure of given organic compound as shown below:
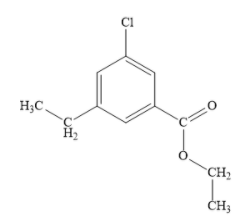
In the above compound, the parent compound is ethyl benzoate. Write the structure of parent compound as shown below:
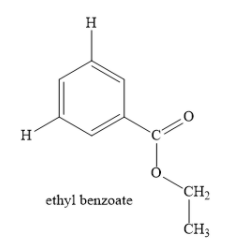
In the parent compound, ethyl ester of benzoic acid is present.
The given compound is the derivative of ethyl benzoate with two substituents on the benzene ring, one is chlorine atom and other is ethyl group.
You can start the numbering of the benzene ring from the carbon atom bearing ester substituent and then go in the direction that gives the lowest locant to the chlorine atom.
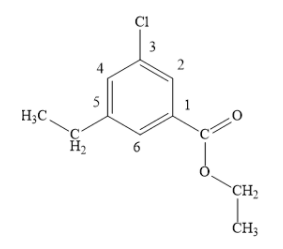
The correct IUPAC name of the given compound is ethyl 3-chloro-5-ethyl benzoate.
Hence, the correct option is the option B.
Note:
Benzoic acid contains a carboxyl group linked to benzene rings. When it reacts with ethanol in presence of sulphuric acid catalyst, ethyl benzoate is obtained.
Complete answer:
The option A is incorrect because it says that the alkyl part of the alcohol from which benzoate easter was obtained, contains chloro substituent which is incorrect.
The option C is incorrect because in the IUPAC system, the prefix meta is not used. Instead the carbon atoms of benzene ring are numbered. So, you can rule out both options C and D.
Draw the structure of given organic compound as shown below:

In the above compound, the parent compound is ethyl benzoate. Write the structure of parent compound as shown below:

In the parent compound, ethyl ester of benzoic acid is present.
The given compound is the derivative of ethyl benzoate with two substituents on the benzene ring, one is chlorine atom and other is ethyl group.
You can start the numbering of the benzene ring from the carbon atom bearing ester substituent and then go in the direction that gives the lowest locant to the chlorine atom.

The correct IUPAC name of the given compound is ethyl 3-chloro-5-ethyl benzoate.
Hence, the correct option is the option B.
Note:
Benzoic acid contains a carboxyl group linked to benzene rings. When it reacts with ethanol in presence of sulphuric acid catalyst, ethyl benzoate is obtained.
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Trending doubts
Which is more stable and why class 12 chemistry CBSE

Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

What are the major means of transport Explain each class 12 social science CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE




