a) i) Explain the preparation of phenol from cumene.
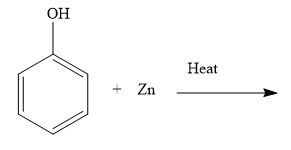
ii) Complete the reaction (please find above image).
b) Explain Williamson's ether synthesis.

Answer
589.2k+ views
Hint:a)
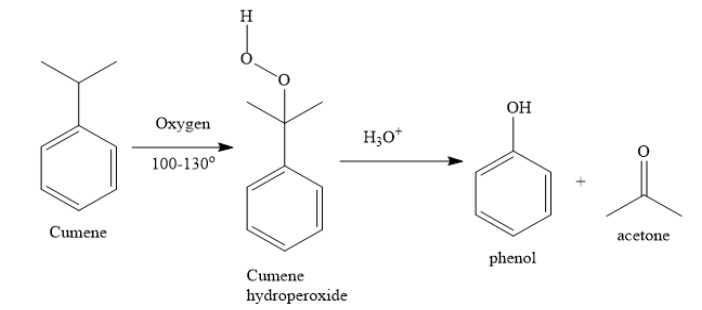
i) Cumene is isopropyl benzene. When you convert cumene to phenol, you need an oxidation reaction.
ii) You exchange an oxygen atom between phenol and zinc.
b) In Williamson ether synthesis, you react to two different substrates to prepare an ether.
Complete answer:
a) i) You can prepare phenol from cumene by air oxidation, followed by acidic hydrolysis. Cumene is isopropyl benzene. Acetone is the side product.
ii) When you heat phenol with zinc, you obtain benzene and zinc oxide. Write the chemical reaction as shown below:
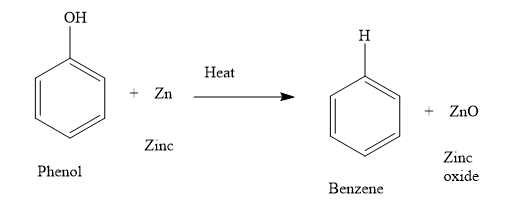
The oxygen atom of phenol is now with zinc. Thus, during the reaction, phenol is reduced and zinc is oxidized. Loss of oxygen is reduction and gain of oxygen is oxidation. The oxidation number of zinc increases from 0 to +2. Increase in the oxidation number is the oxidation and decrease in the oxidation number is reduction.
b) In the Williamson ether synthesis, you prepare an ether from organo halides and a deprotonated alcohol (alkoxide).

Thus, you react sodium ethoxide with ethyl chloride to obtain diethyl ether. Sodium chloride is the by-product.
Note:
a) i) Apart from the preparation of phenol from cumene, there are various other methods by which you can prepare phenol.
ii) In the redox reaction, one substance is reduced and acts as an oxidizing agent. The other substance is oxidized and acts as a reducing agent.
b) With the help of Williamson's ether synthesis, you can prepare simple ethers and mixed ethers. However the purification of mixed ethers is complicated.
i) Cumene is isopropyl benzene. When you convert cumene to phenol, you need an oxidation reaction.
ii) You exchange an oxygen atom between phenol and zinc.
b) In Williamson ether synthesis, you react to two different substrates to prepare an ether.
Complete answer:
a) i) You can prepare phenol from cumene by air oxidation, followed by acidic hydrolysis. Cumene is isopropyl benzene. Acetone is the side product.

ii) When you heat phenol with zinc, you obtain benzene and zinc oxide. Write the chemical reaction as shown below:

The oxygen atom of phenol is now with zinc. Thus, during the reaction, phenol is reduced and zinc is oxidized. Loss of oxygen is reduction and gain of oxygen is oxidation. The oxidation number of zinc increases from 0 to +2. Increase in the oxidation number is the oxidation and decrease in the oxidation number is reduction.
b) In the Williamson ether synthesis, you prepare an ether from organo halides and a deprotonated alcohol (alkoxide).

Thus, you react sodium ethoxide with ethyl chloride to obtain diethyl ether. Sodium chloride is the by-product.
Note:
a) i) Apart from the preparation of phenol from cumene, there are various other methods by which you can prepare phenol.
ii) In the redox reaction, one substance is reduced and acts as an oxidizing agent. The other substance is oxidized and acts as a reducing agent.
b) With the help of Williamson's ether synthesis, you can prepare simple ethers and mixed ethers. However the purification of mixed ethers is complicated.
Recently Updated Pages
Three beakers labelled as A B and C each containing 25 mL of water were taken A small amount of NaOH anhydrous CuSO4 and NaCl were added to the beakers A B and C respectively It was observed that there was an increase in the temperature of the solutions contained in beakers A and B whereas in case of beaker C the temperature of the solution falls Which one of the following statements isarecorrect i In beakers A and B exothermic process has occurred ii In beakers A and B endothermic process has occurred iii In beaker C exothermic process has occurred iv In beaker C endothermic process has occurred

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

What are the major means of transport Explain each class 12 social science CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE

Why should a magnesium ribbon be cleaned before burning class 12 chemistry CBSE




