A hydrocarbon of molecular formula ${{C}_{5}}{{H}_{10}}$ on monochlorination gives one product and on chlorination gives three products (excluding the stereoisomers). Identify the hydrocarbon.
Answer
621.6k+ views
Hint: It is an alicyclic hydrocarbon and a highly flammable compound. This compound is made by the cracking of cyclohexane in the presence of high temperature and pressure.
Complete step by step answer:
- This question is done basically by a hit and trial method.
- From the first part of the question which is the molecular formula, ${{C}_{5}}{{H}_{10}}$ comes under the general formula of ${{C}_{n}}{{H}_{2n}}$.
- From here we can get to know that the compound is either an alkene or a cyclic compound.
- So, the possible molecules are pentene and cyclopentane.
- From this point, we can look at the second part of the question, which says that the monochlorination of this compound should give only one product.
- In case of pentene (whether it is 1-pentene or 2-pentene), there are two $\alpha $-hydrogens and so, on monochlorination, there will be two different products formed.

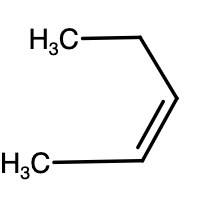
- Above given are the structures of pentene. The reaction is given as,
\[C{{H}_{2}}=CHC{{H}_{2}}C{{H}_{2}}C{{H}_{3}}+HCl\to Cl-C{{H}_{2}}-C{{H}_{2}}C{{H}_{2}}C{{H}_{2}}C{{H}_{3}}+C{{H}_{3}}-\underset{\underset{Cl}{\mathop{|}}\,}{\mathop{CH}}\,-C{{H}_{2}}C{{H}_{2}}C{{H}_{3}}\]
-As we can see above, monochlorination of 1-pentene results in two different products. Similar reaction will take place for 2-pentene as well.
- So, we can now check the monochlorination product of cyclopentane.
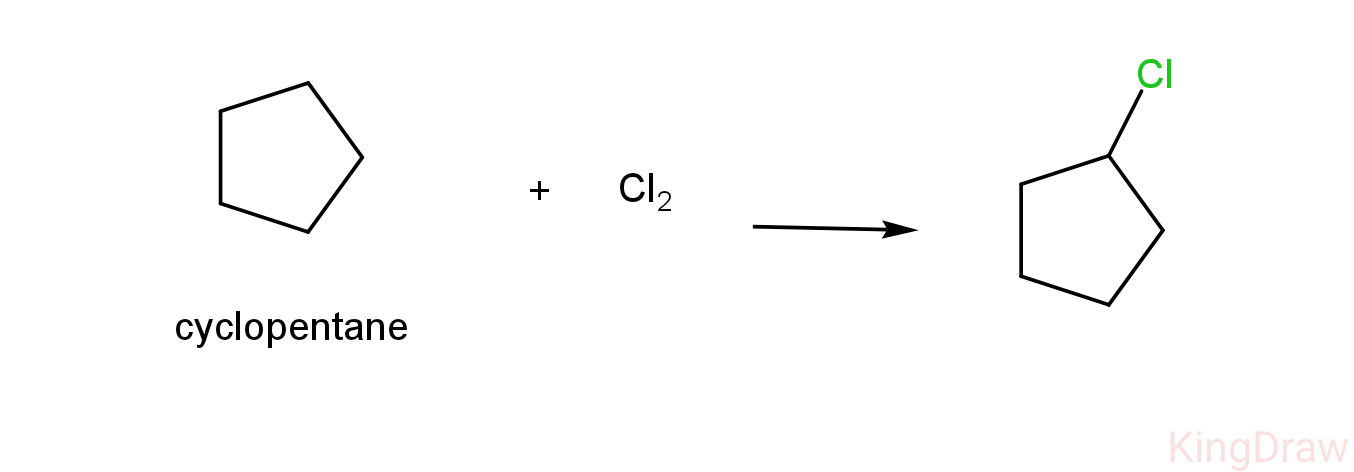
-As we can see above, cyclopentane reacts with chlorine to form only one product which is 1-chlorocyclopentane.
- Now, to confirm the compound, we can see the next part of the question which says that on dichlorination it should give three products
- Given below is the dichlorination of cyclopentane.
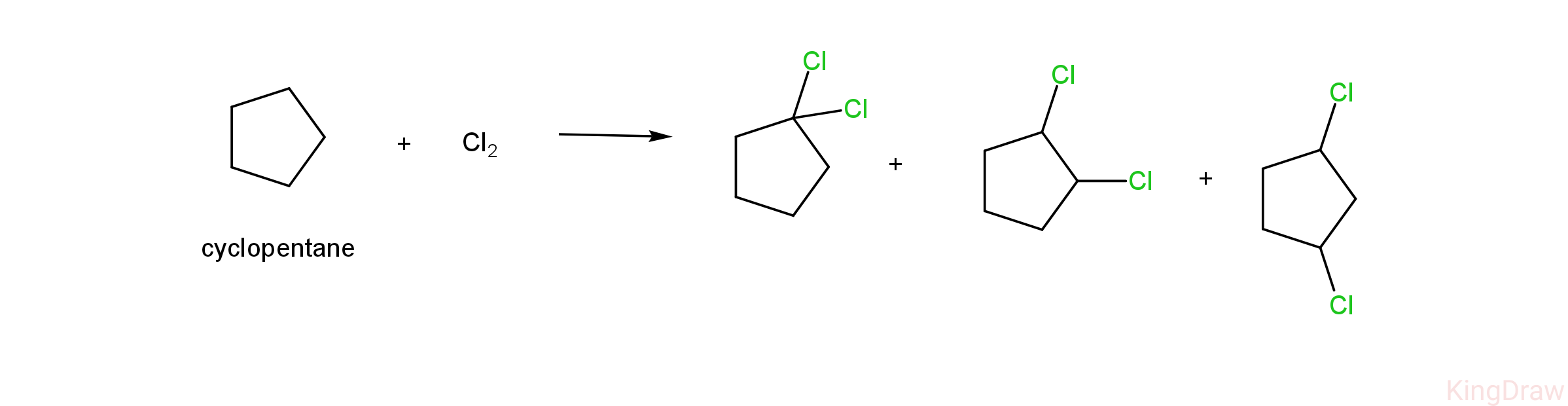
- As we can see, the dichlorination of cyclopentane results in the formation of three products, which are 1,1-dichlorocyclopentane, 1,2 -dichlorocyclopentane and 1,3-dichlorocyclopentane.
- Therefore , we can conclude that cyclopentane is the compound having the molecular formula of C5H10, on monochlorination giving one product (1-chloro cyclopentane) and on dichlorination gives three products (1,1-dichloro cyclopentane, 1,2 -dichlorocyclopentane and 1,3- dichlorocyclopentane)
- So, the answer is Cyclopentane.
Note: Monochlorination and dichlorination both the reaction proceeds under different sets of conditions. Monochlorination of cyclopentane occurs with $C{{l}_{2}}$ in the presence of light ($h\upsilon $) whereas dichlorination of cyclopentene occurs when excess of $C{{l}_{2}}$ is provided in the presence of light ($h\upsilon $).
Complete step by step answer:
- This question is done basically by a hit and trial method.
- From the first part of the question which is the molecular formula, ${{C}_{5}}{{H}_{10}}$ comes under the general formula of ${{C}_{n}}{{H}_{2n}}$.
- From here we can get to know that the compound is either an alkene or a cyclic compound.
- So, the possible molecules are pentene and cyclopentane.
- From this point, we can look at the second part of the question, which says that the monochlorination of this compound should give only one product.
- In case of pentene (whether it is 1-pentene or 2-pentene), there are two $\alpha $-hydrogens and so, on monochlorination, there will be two different products formed.


- Above given are the structures of pentene. The reaction is given as,
\[C{{H}_{2}}=CHC{{H}_{2}}C{{H}_{2}}C{{H}_{3}}+HCl\to Cl-C{{H}_{2}}-C{{H}_{2}}C{{H}_{2}}C{{H}_{2}}C{{H}_{3}}+C{{H}_{3}}-\underset{\underset{Cl}{\mathop{|}}\,}{\mathop{CH}}\,-C{{H}_{2}}C{{H}_{2}}C{{H}_{3}}\]
-As we can see above, monochlorination of 1-pentene results in two different products. Similar reaction will take place for 2-pentene as well.
- So, we can now check the monochlorination product of cyclopentane.


-As we can see above, cyclopentane reacts with chlorine to form only one product which is 1-chlorocyclopentane.
- Now, to confirm the compound, we can see the next part of the question which says that on dichlorination it should give three products
- Given below is the dichlorination of cyclopentane.

- As we can see, the dichlorination of cyclopentane results in the formation of three products, which are 1,1-dichlorocyclopentane, 1,2 -dichlorocyclopentane and 1,3-dichlorocyclopentane.
- Therefore , we can conclude that cyclopentane is the compound having the molecular formula of C5H10, on monochlorination giving one product (1-chloro cyclopentane) and on dichlorination gives three products (1,1-dichloro cyclopentane, 1,2 -dichlorocyclopentane and 1,3- dichlorocyclopentane)
- So, the answer is Cyclopentane.
Note: Monochlorination and dichlorination both the reaction proceeds under different sets of conditions. Monochlorination of cyclopentane occurs with $C{{l}_{2}}$ in the presence of light ($h\upsilon $) whereas dichlorination of cyclopentene occurs when excess of $C{{l}_{2}}$ is provided in the presence of light ($h\upsilon $).
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Social Science: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

Why is the cell called the structural and functional class 12 biology CBSE

Draw ray diagrams each showing i myopic eye and ii class 12 physics CBSE

Which is the correct genotypic ratio of mendel dihybrid class 12 biology CBSE




